The "Other" Inositols and Their Phosphates: Synthesis, Biology, and Medicine (with Recent Advances in myo-Inositol Chemistry)
- PMID: 26694856
- PMCID: PMC5156312
- DOI: 10.1002/anie.201502227
The "Other" Inositols and Their Phosphates: Synthesis, Biology, and Medicine (with Recent Advances in myo-Inositol Chemistry)
Abstract
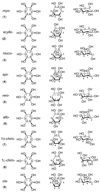
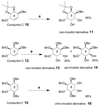
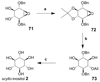
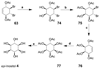
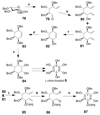
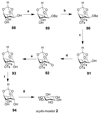
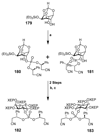
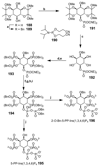
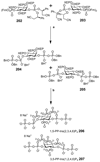
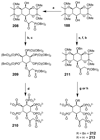
Cell signaling via inositol phosphates, in particular via the second messenger myo-inositol 1,4,5-trisphosphate, and phosphoinositides comprises a huge field of biology. Of the nine 1,2,3,4,5,6-cyclohexanehexol isomers, myo-inositol is pre-eminent, with "other" inositols (cis-, epi-, allo-, muco-, neo-, L-chiro-, D-chiro-, and scyllo-) and derivatives rarer or thought not to exist in nature. However, neo- and d-chiro-inositol hexakisphosphates were recently revealed in both terrestrial and aquatic ecosystems, thus highlighting the paucity of knowledge of the origins and potential biological functions of such stereoisomers, a prevalent group of environmental organic phosphates, and their parent inositols. Some "other" inositols are medically relevant, for example, scyllo-inositol (neurodegenerative diseases) and d-chiro-inositol (diabetes). It is timely to consider exploration of the roles and applications of the "other" isomers and their derivatives, likely by exploiting techniques now well developed for the myo series.
Keywords: cyclitols; inositol; isomers; phosphates; synthetic methods.
© 2016 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures





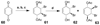

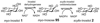












References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

