Drug-Induced Liver Injury: Pattern Recognition and Future Directions
- PMID: 26696029
- PMCID: PMC4694731
- DOI: 10.5009/gnl15114
Drug-Induced Liver Injury: Pattern Recognition and Future Directions
Abstract
Drug-induced liver injury (DILI) remains a significant clinical challenge and is the leading cause of acute liver failure in most countries. An aging population that uses more medications, a constant influx of newly developed drugs and a growing risk from unfamiliar herbal and dietary supplements will make DILI an increasing part of clinical practice. Currently, the most effective strategy for disease management is rapid identification, withholding the inciting agents, supportive care and having a firm understanding of the expected natural history. There are resources available to aid the clinician, including a new online "textbook" as well as causality assessment tools, but a heightened awareness of risk and the disease's varying phenotypes and good history-taking remain cornerstones to diagnosis. Looking ahead, growing registries of cases, pharmacoepidemiology studies and translational research into the mechanisms of injury may produce better diagnostic tools, markers for risk and disease, and prevention and therapeutics.
Keywords: Diagnosis; Drug induced liver injury; Epidemiology; Hepatotoxicity; Herbal and dietary supplements.
Figures
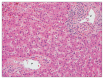

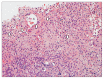
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

