Human iPSC for Therapeutic Approaches to the Nervous System: Present and Future Applications
- PMID: 26697076
- PMCID: PMC4677260
- DOI: 10.1155/2016/4869071
Human iPSC for Therapeutic Approaches to the Nervous System: Present and Future Applications
Abstract
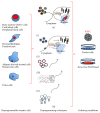
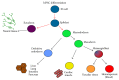
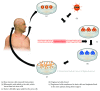
Many central nervous system (CNS) diseases including stroke, spinal cord injury (SCI), and brain tumors are a significant cause of worldwide morbidity/mortality and yet do not have satisfying treatments. Cell-based therapy to restore lost function or to carry new therapeutic genes is a promising new therapeutic approach, particularly after human iPSCs became available. However, efficient generation of footprint-free and xeno-free human iPSC is a prerequisite for their clinical use. In this paper, we will first summarize the current methodology to obtain footprint- and xeno-free human iPSC. We will then review the current iPSC applications in therapeutic approaches for CNS regeneration and their use as vectors to carry proapoptotic genes for brain tumors and review their applications for modelling of neurological diseases and formulating new therapeutic approaches. Available results will be summarized and compared. Finally, we will discuss current limitations precluding iPSC from being used on large scale for clinical applications and provide an overview of future areas of improvement. In conclusion, significant progress has occurred in deriving iPSC suitable for clinical use in the field of neurological diseases. Current efforts to overcome technical challenges, including reducing labour and cost, will hopefully expedite the integration of this technology in the clinical setting.
Figures

Similar articles
-
Footprint- and xeno-free human iPSCs derived from urine cells using extracellular matrix-based culture conditions.Biomaterials. 2014 Sep;35(29):8330-8. doi: 10.1016/j.biomaterials.2014.05.059. Epub 2014 Jun 30. Biomaterials. 2014. PMID: 24994040
-
Applications of induced pluripotent stem cell technologies in spinal cord injury.J Neurochem. 2017 Jun;141(6):848-860. doi: 10.1111/jnc.13986. Epub 2017 Apr 5. J Neurochem. 2017. PMID: 28199003 Review.
-
Induced pluripotent stem cells, a giant leap for mankind therapeutic applications.World J Stem Cells. 2019 Jul 26;11(7):421-430. doi: 10.4252/wjsc.v11.i7.421. World J Stem Cells. 2019. PMID: 31396369 Free PMC article. Review.
-
[Therapeutic potential of induced pluripotent stem cells for spinal cord injury].Brain Nerve. 2012 Jan;64(1):17-27. Brain Nerve. 2012. PMID: 22223498 Review. Japanese.
-
Recent Progress in the Regeneration of Spinal Cord Injuries by Induced Pluripotent Stem Cells.Int J Mol Sci. 2019 Aug 6;20(15):3838. doi: 10.3390/ijms20153838. Int J Mol Sci. 2019. PMID: 31390782 Free PMC article. Review.
Cited by
-
Synergistic and Superimposed Effect of Bone Marrow-Derived Mesenchymal Stem Cells Combined with Fasudil in Experimental Autoimmune Encephalomyelitis.J Mol Neurosci. 2016 Dec;60(4):486-497. doi: 10.1007/s12031-016-0819-3. Epub 2016 Aug 30. J Mol Neurosci. 2016. PMID: 27573128
-
Tissue Engineered Neural Constructs Composed of Neural Precursor Cells, Recombinant Spidroin and PRP for Neural Tissue Regeneration.Sci Rep. 2019 Feb 28;9(1):3161. doi: 10.1038/s41598-019-39341-9. Sci Rep. 2019. PMID: 30816182 Free PMC article.
-
Transplanting neural progenitor cells to restore connectivity after spinal cord injury.Nat Rev Neurosci. 2020 Jul;21(7):366-383. doi: 10.1038/s41583-020-0314-2. Epub 2020 Jun 9. Nat Rev Neurosci. 2020. PMID: 32518349 Free PMC article. Review.
-
Use of Brain-Derived Stem/Progenitor Cells and Derived Extracellular Vesicles to Repair Damaged Neural Tissues: Lessons Learned from Connective Tissue Repair Regarding Variables Limiting Progress and Approaches to Overcome Limitations.Int J Mol Sci. 2023 Feb 8;24(4):3370. doi: 10.3390/ijms24043370. Int J Mol Sci. 2023. PMID: 36834779 Free PMC article. Review.
-
The Application of Brain Organoid Technology in Stroke Research: Challenges and Prospects.Front Cell Neurosci. 2021 Jun 21;15:646921. doi: 10.3389/fncel.2021.646921. eCollection 2021. Front Cell Neurosci. 2021. PMID: 34234646 Free PMC article. Review.
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

