Electrolytic extraction drives volatile fatty acid chain elongation through lactic acid and replaces chemical pH control in thin stillage fermentation
- PMID: 26697110
- PMCID: PMC4687354
- DOI: 10.1186/s13068-015-0396-7
Electrolytic extraction drives volatile fatty acid chain elongation through lactic acid and replaces chemical pH control in thin stillage fermentation
Abstract
Background: Volatile fatty acids (VFA) are building blocks for the chemical industry. Sustainable, biological production is constrained by production and recovery costs, including the need for intensive pH correction. Membrane electrolysis has been developed as an in situ extraction technology tailored to the direct recovery of VFA from fermentation while stabilizing acidogenesis without caustic addition. A current applied across an anion exchange membrane reduces the fermentation broth (catholyte, water reduction: H2O + e(-) → ½ H2 + OH(-)) and drives carboxylate ions into a clean, concentrated VFA stream (anolyte, water oxidation: H2O → 2e(-) + 2 H(+) + O2).
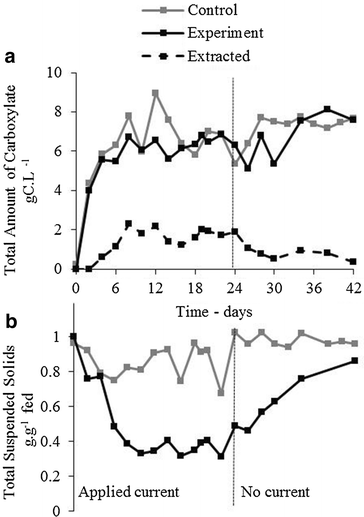
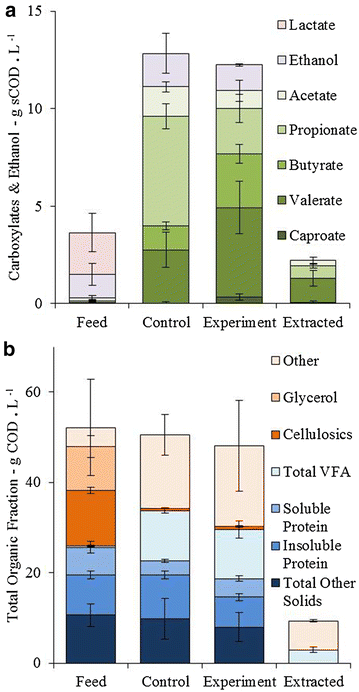
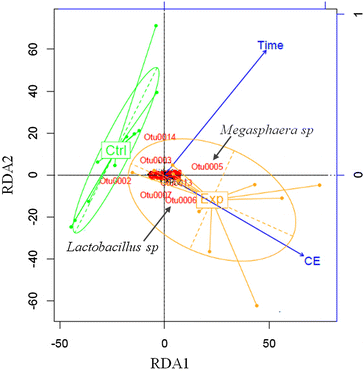
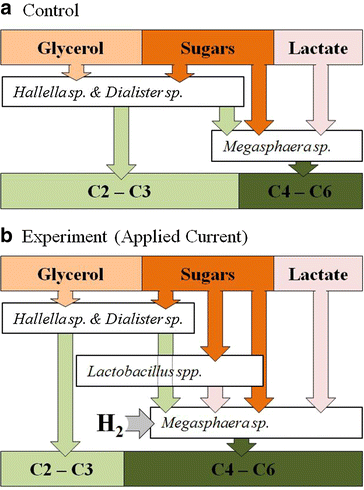
Results: In this study, we fermented thin stillage to generate a mixed VFA extract without chemical pH control. Membrane electrolysis (0.1 A, 3.22 ± 0.60 V) extracted 28 ± 6 % of carboxylates generated per day (on a carbon basis) and completely replaced caustic control of pH, with no impact on the total carboxylate production amount or rate. Hydrogen generated from the applied current shifted the fermentation outcome from predominantly C2 and C3 VFA (64 ± 3 % of the total VFA present in the control) to majority of C4 to C6 (70 ± 12 % in the experiment), with identical proportions in the VFA acid extract. A strain related to Megasphaera elsdenii (maximum abundance of 57 %), a bacteria capable of producing mid-chain VFA at a high rate, was enriched by the applied current, alongside a stable community of Lactobacillus spp. (10 %), enabling chain elongation of VFA through lactic acid. A conversion of 30 ± 5 % VFA produced per sCOD fed (60 ± 10 % of the reactive fraction) was achieved, with a 50 ± 6 % reduction in suspended solids likely by electro-coagulation.
Conclusions: VFA can be extracted directly from a fermentation broth by membrane electrolysis. The electrolytic water reduction products are utilized in the fermentation: OH(-) is used for pH control without added chemicals, and H2 is metabolized by species such as Megasphaera elsdenii to produce greater value, more reduced VFA. Electro-fermentation displays promise for generating added value chemical co-products from biorefinery sidestreams and wastes.
Keywords: Biorefinery; Carboxylate platform; Chain elongation; Electro-fermentation; Extraction; Membrane electrolysis.
Figures

Similar articles
-
Electrolytic membrane extraction enables production of fine chemicals from biorefinery sidestreams.Environ Sci Technol. 2014 Jun 17;48(12):7135-42. doi: 10.1021/es500483w. Epub 2014 Jun 3. Environ Sci Technol. 2014. PMID: 24844669
-
Fermentation of alfalfa wet-fractionation liquids to volatile fatty acids by Streptococcus bovis and Megasphaera elsdenii.Bioresour Technol. 2013 Aug;142:88-94. doi: 10.1016/j.biortech.2013.05.016. Epub 2013 May 14. Bioresour Technol. 2013. PMID: 23732922
-
pH-adjustment strategy for volatile fatty acid production from high-strength wastewater for biological nutrient removal.Water Sci Technol. 2014;69(10):2043-51. doi: 10.2166/wst.2014.120. Water Sci Technol. 2014. PMID: 24845319
-
Upflow anaerobic sludge blanket reactor--a review.Indian J Environ Health. 2001 Apr;43(2):1-82. Indian J Environ Health. 2001. PMID: 12397675 Review.
-
Bio-based volatile fatty acid production and recovery from waste streams: Current status and future challenges.Bioresour Technol. 2018 Nov;268:773-786. doi: 10.1016/j.biortech.2018.07.042. Epub 2018 Jul 11. Bioresour Technol. 2018. PMID: 30030049 Review.
Cited by
-
Increasing the economic value of lignocellulosic stillage through medium-chain fatty acid production.Biotechnol Biofuels. 2018 Jul 19;11:200. doi: 10.1186/s13068-018-1193-x. eCollection 2018. Biotechnol Biofuels. 2018. PMID: 30034526 Free PMC article.
-
Trophic interactions shape the spatial organization of medium-chain carboxylic acid producing granular biofilm communities.ISME J. 2023 Nov;17(11):2014-2022. doi: 10.1038/s41396-023-01508-8. Epub 2023 Sep 15. ISME J. 2023. PMID: 37715042 Free PMC article.
-
Electricity-assisted production of caproic acid from grass.Biotechnol Biofuels. 2017 Jul 11;10:180. doi: 10.1186/s13068-017-0863-4. eCollection 2017. Biotechnol Biofuels. 2017. PMID: 28702085 Free PMC article.
-
Evaluation of a Prototype for Electrochemical pH-Shift Crystallization of Succinic Acid.Materials (Basel). 2022 Nov 25;15(23):8412. doi: 10.3390/ma15238412. Materials (Basel). 2022. PMID: 36499913 Free PMC article.
-
Selective Extraction of Medium-Chain Carboxylic Acids by Electrodialysis and Phase Separation.ACS Omega. 2021 Mar 12;6(11):7841-7850. doi: 10.1021/acsomega.1c00397. eCollection 2021 Mar 23. ACS Omega. 2021. PMID: 33778296 Free PMC article.
References
-
- Deloitte. Opportunities for the fermentation-based chemical industry: an analysis of the market potential and competitiveness of North-West Europe. September 2014; http://www2.deloitte.com/global/en/pages/manufacturing/articles/opportun.... Accessed 4th Aug 2015.
-
- Wagemann K. Herstellung von Grundchemikalien auf Basis nachwachsender Rohstoffe als Alternative zur Petrochemie?/Production of basic chemicals on the basis of renewable resources as alternative to petrochemistry? Chelie Ingenieur Technik. 2014;86(12):2115–2134. doi: 10.1002/cite.201400108. - DOI
-
- Ramaswamy S, Huang HJ, Ramarao BV. Separation and purification technologies in biorefineries. Hoboken: Wiley. 2013. doi:10.1002/9781118493441.
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

