Galvanic Corrosion of and Ion Release from Various Orthodontic Brackets and Wires in a Fluoride-containing Mouthwash
- PMID: 26697148
- PMCID: PMC4682012
- DOI: 10.15171/joddd.2015.030
Galvanic Corrosion of and Ion Release from Various Orthodontic Brackets and Wires in a Fluoride-containing Mouthwash
Abstract
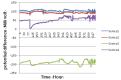
Background and aims. This study compared the galvanic corrosion of orthodontic wires and brackets from various manufacturers following exposure to a fluoride mouthwash. Materials and methods. This study was conducted on 24 lower central incisor 0.022" Roth brackets of four different commercially available brands (Dentaurum, American Orthodontics, ORJ, Shinye). These brackets along with stainless steel (SS) or nickel-titanium (NiTi) orthodontic wires (0.016", round) were immersed in Oral-B mouthwash containing 0.05% sodium fluoride for 28 days. The electric potential (EP) difference of each bracket-wire couple was measured with a Saturated Calomel Reference Electrode (Ag/AgCl saturated with KCl) via a voltmeter. The ions released in the electrolyte weremeasured with an atomic absorption spectrometer. All the specimens were assessed under a stereomicroscope and specimens with corrosion were analyzed with scanning electron microscopy (SEM). Data were analyzed using ANOVA. Results. The copper ions released from specimens with NiTi wire were greater than those of samples containing SS wire. ORJ brackets released more Cu ions than other samples. The Ni ions released from Shinye brackets were significantly more than those of other specimens (P < 0.05). Corrosion rate of brackets coupled with NiTi wires was higher than that of brackets coupled with SS wires. Light and electron microscopic observations showed greater corrosion of ORJ brackets. Conclusion. In fluoride mouthwash, Shinye and ORJ brackets exhibited greater corrosion than Dentaurum and American Orthodontics brackets. Stainless steel brackets used with NiTi wires showed greater corrosion and thus caution is recommended when using them.
Keywords: Corrosion; nickel; orthodontic bracket; sodium fluoride.
Similar articles
-
Galvanic coupling of steel and gold alloy lingual brackets with orthodontic wires: Is corrosion a concern?Angle Orthod. 2018 Jul;88(4):450-457. doi: 10.2319/092917-655.1. Epub 2018 Mar 6. Angle Orthod. 2018. PMID: 29509025 Free PMC article.
-
Ion Release and Galvanic Corrosion of Different Orthodontic Brackets and Wires in Artificial Saliva.J Contemp Dent Pract. 2017 Mar 1;18(3):222-227. J Contemp Dent Pract. 2017. PMID: 28258269
-
Galvanic corrosion between orthodontic wires and brackets in fluoride mouthwashes.Eur J Orthod. 2006 Jun;28(3):298-304. doi: 10.1093/ejo/cji102. Epub 2006 Jan 20. Eur J Orthod. 2006. PMID: 16428255
-
Comparison of galvanic corrosion potential of metal injection molded brackets to that of conventional metal brackets with nickel-titanium and copper nickel-titanium archwire combinations.J Contemp Dent Pract. 2013 May 1;14(3):488-95. doi: 10.5005/jp-journals-10024-1350. J Contemp Dent Pract. 2013. PMID: 24171995
-
Effect of Fluoridated Mouthwashes on Corrosion Property of Orthodontic Appliances: A Narrative Review.J Contemp Dent Pract. 2022 Apr 1;23(4):460-466. J Contemp Dent Pract. 2022. PMID: 35945842 Review.
Cited by
-
Galvanic coupling of steel and gold alloy lingual brackets with orthodontic wires: Is corrosion a concern?Angle Orthod. 2018 Jul;88(4):450-457. doi: 10.2319/092917-655.1. Epub 2018 Mar 6. Angle Orthod. 2018. PMID: 29509025 Free PMC article.
-
Fluorinated Agents Effects on Orthodontic Alloys: A Descriptive In Vitro Study.Materials (Basel). 2022 Jun 30;15(13):4612. doi: 10.3390/ma15134612. Materials (Basel). 2022. PMID: 35806740 Free PMC article.
-
Metal Ion Release from Orthodontic Archwires: A Comparative Study of Biocompatibility and Corrosion Resistance.Molecules. 2024 Nov 30;29(23):5685. doi: 10.3390/molecules29235685. Molecules. 2024. PMID: 39683842 Free PMC article.
-
Nickel and Chromium Ion Release from Orthodontic Wires Subjected to Various Drinks and Distilled Water.Front Dent. 2023 Sep 18;20:33. doi: 10.18502/fid.v20i33.13639. eCollection 2023. Front Dent. 2023. PMID: 37876792 Free PMC article.
-
Effect of arch wires and brackets in orthodontics for releasing nickel ions.Bioinformation. 2025 Jan 31;21(1):35-39. doi: 10.6026/973206300210035. eCollection 2025. Bioinformation. 2025. PMID: 40255301 Free PMC article.
References
-
- Iijima M, Endo K, Yuasa T, Ohno H, Hayashi K, Kakizaki M. et al. Galvanic corrosion behavior of orthodontic archwire alloys coupled to bracket alloys. Angle Orthod. 2006;76:705–11. - PubMed
-
- Jahanbin A, Shahabi M, Mokhber N, Tavakkolian Ardakani E. Comparison of nickel ion release and corrosion sites among commonly used stainless steel brackets in Iran. J Mash Dent Sch. 2009;33:17–24.
-
- Kolokitha OG, Chatzistavrou E. Allergic reactions to nickel-containing orthodontic appliances: clinical signs and treatment alternatives. World J Orthod. 2008;9:399–406. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources