Cardioprotection and Safety of Dexrazoxane in Patients Treated for Newly Diagnosed T-Cell Acute Lymphoblastic Leukemia or Advanced-Stage Lymphoblastic Non-Hodgkin Lymphoma: A Report of the Children's Oncology Group Randomized Trial Pediatric Oncology Group 9404
- PMID: 26700126
- PMCID: PMC4872007
- DOI: 10.1200/JCO.2015.60.8851
Cardioprotection and Safety of Dexrazoxane in Patients Treated for Newly Diagnosed T-Cell Acute Lymphoblastic Leukemia or Advanced-Stage Lymphoblastic Non-Hodgkin Lymphoma: A Report of the Children's Oncology Group Randomized Trial Pediatric Oncology Group 9404
Erratum in
-
Errata.J Clin Oncol. 2017 Jun 20;35(18):2100. doi: 10.1200/JCO.2017.73.9433. J Clin Oncol. 2017. PMID: 28618244 Free PMC article. No abstract available.
Abstract
Purpose: To determine the oncologic efficacy, cardioprotective effectiveness, and safety of dexrazoxane added to chemotherapy that included a cumulative doxorubicin dose of 360 mg/m(2) to treat children and adolescents with newly diagnosed T-cell acute lymphoblastic leukemia (T-ALL) or lymphoblastic non-Hodgkin lymphoma (L-NHL).
Patients and methods: Patients were treated on Pediatric Oncology Group Protocol POG 9404, which included random assignment to treatment with or without dexrazoxane given as a bolus infusion immediately before every dose of doxorubicin. Cardiac effects were assessed by echocardiographic measurements of left ventricular function and structure.
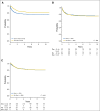
Results: Of 573 enrolled patients, 537 were eligible, evaluable, and randomly assigned to an arm with or without dexrazoxane. The 5-year event-free survival (with standard error) did not differ between groups: 77.2% (2.7%) for the dexrazoxane group versus 76.0% (2.7%) for the doxorubicin-only group (P = .9). The frequencies of severe grade 3 or 4 hematologic toxicity, infection, CNS events, and toxic deaths were similar in both groups (P ranged from .26 to .64). Of 11 second malignancies, eight occurred in patients who received dexrazoxane (P = .17). The mean left ventricular fractional shortening, wall thickness, and thickness-to-dimension ratio z scores measured 3 years after diagnosis were worse in the doxorubicin-alone group (n = 55 per group; P ≤ .01 for all comparisons). Mean fractional shortening z scores measured 3.5 to 6.4 years after diagnosis remained diminished and were lower in the 21 patients who received doxorubicin alone than in the 31 patients who received dexrazoxane (-2.03 v -0.24; P ≤ .001).
Conclusion: Dexrazoxane was cardioprotective and did not compromise antitumor efficacy, did not increase the frequencies of toxicities, and was not associated with a significant increase in second malignancies with this doxorubicin-containing chemotherapy regimen. We recommend dexrazoxane as a cardioprotectant for children and adolescents who have malignancies treated with anthracyclines.
© 2015 by American Society of Clinical Oncology.
Conflict of interest statement
Authors' disclosures of potential conflicts of interest are found in the article online at www.jco.org. Author contributions are found at the end of this article.
Figures



References
-
- Reiter A, Schrappe M, Ludwig WD, et al. Chemotherapy in 998 unselected childhood acute lymphoblastic leukemia patients: Results and conclusions of the multicenter trial ALL-BFM 86. Blood. 1994;84:3122–3133. - PubMed
-
- Schrappe M, Reiter A, Ludwig WD, et al. German-Austrian-Swiss ALL-BFM Study Group Improved outcome in childhood acute lymphoblastic leukemia despite reduced use of anthracyclines and cranial radiotherapy: Results of trial ALL-BFM 90. Blood. 2000;95:3310–3322. - PubMed
-
- Goldberg JM, Silverman LB, Levy DE, et al. Childhood T-cell acute lymphoblastic leukemia: The Dana-Farber Cancer Institute acute lymphoblastic leukemia consortium experience. J Clin Oncol. 2003;21:3616–3622. - PubMed
-
- Oeffinger KC, Mertens AC, Sklar CA, et al. Childhood Cancer Survivor Study Chronic health conditions in adult survivors of childhood cancer. N Engl J Med. 2006;355:1572–1582. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

