Identification and Functional Characterization of Glycosylation of Recombinant Human Platelet-Derived Growth Factor-BB in Pichia pastoris
- PMID: 26701617
- PMCID: PMC4689512
- DOI: 10.1371/journal.pone.0145419
Identification and Functional Characterization of Glycosylation of Recombinant Human Platelet-Derived Growth Factor-BB in Pichia pastoris
Abstract
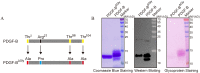
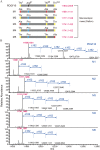
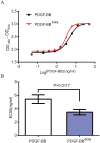
Yeast Pichia pastoris is a widely used system for heterologous protein expression. However, post-translational modifications, especially glycosylation, usually impede pharmaceutical application of recombinant proteins because of unexpected alterations in protein structure and function. The aim of this study was to identify glycosylation sites on recombinant human platelet-derived growth factor-BB (rhPDGF-BB) secreted by P. pastoris, and investigate possible effects of O-linked glycans on PDGF-BB functional activity. PDGF-BB secreted by P. pastoris is very heterogeneous and contains multiple isoforms. We demonstrated that PDGF-BB was O-glycosylated during the secretion process and detected putative O-glycosylation sites using glycosylation staining and immunoblotting. By site-directed mutagenesis and high-resolution LC/MS analysis, we, for the first time, identified two threonine residues at the C-terminus as the major O-glycosylation sites on rhPDGF-BB produced in P. pastoris. Although O-glycosylation resulted in heterogeneous protein expression, the removal of glycosylation sites did not affect rhPDGF-BB mitogenic activity. In addition, the unglycosylated PDGF-BBΔGly mutant exhibited the immunogenicity comparable to that of the wild-type form. Furthermore, antiserum against PDGF-BBΔGly also recognized glycosylated PDGF-BB, indicating that protein immunogenicity was unaltered by glycosylation. These findings elucidate the effect of glycosylation on PDGF-BB structure and biological activity, and can potentially contribute to the design and production of homogeneously expressed unglycosylated or human-type glycosylated PDGF-BB in P. pastoris for pharmaceutical applications.
Conflict of interest statement
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

