Sclerostin Immunoreactivity Increases in Cortical Bone Osteocytes and Decreases in Articular Cartilage Chondrocytes in Aging Mice
- PMID: 26701970
- PMCID: PMC4810796
- DOI: 10.1369/0022155415626499
Sclerostin Immunoreactivity Increases in Cortical Bone Osteocytes and Decreases in Articular Cartilage Chondrocytes in Aging Mice
Abstract
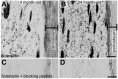
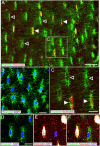
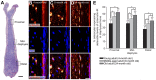
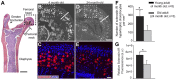
Sclerostin is a 24-kDa secreted glycoprotein that has been identified as a negative modulator of new bone formation and may play a major role in age-related decline in skeletal function. Although serum levels of sclerostin markedly increase with age, relatively little is known about whether cells in the skeleton change their expression of sclerostin with aging. Using immunohistochemistry and confocal microscopy, we explored sclerostin immunoreactivity (sclerostin-IR) in the femurs of 4-, 9-, and 24-month-old adult C3H/HeJ male mice. In the femur, the only two cell types that expressed detectable levels of sclerostin-IR were bone osteocytes and articular cartilage chondrocytes. At three different sites along the diaphysis of the femur, only a subset of osteocytes expressed sclerostin-IR and the percentage of osteocytes that expressed sclerostin-IR increased from approximately 36% to 48% in 4- vs. 24-month-old mice. In marked contrast, in the same femurs, there were ~40% fewer hypertrophic chondrocytes of articular cartilage that expressed sclerostin-IR when comparing 24- vs. 4-month-old mice. Understanding the mechanism(s) that drive these divergent changes in sclerostin-IR may provide insight into understanding and treating the age-related decline of the skeleton.
Keywords: aged; articular cartilage; cortical bone; hypertrophic chondrocyte; joint; osteocyte subtype.
© 2016 The Histochemical Society.
Conflict of interest statement
Figures

References
-
- Al-Qtaitat A, Shore RC, Aaron JE. (2010). Structural changes in the ageing periosteum using collagen III immuno-staining and chromium labelling as indicators. J Musculoskelet Neuronal Interact 10:112-123. - PubMed
-
- Allen MR, Burr DB. (2011). Bisphosphonate effects on bone turnover, microdamage, and mechanical properties: what we think we know and what we know that we don’t know. Bone 49:56-65. - PubMed
-
- Almeida M, Han L, Martin-Millan M, Plotkin LI, Stewart SA, Roberson PK, Kousteni S, O’Brien CA, Bellido T, Parfitt AM, Weinstein RS, Jilka RL, Manolagas SC. (2007). Skeletal involution by age-associated oxidative stress and its acceleration by loss of sex steroids. J Biol Chem 282:27285-27297. - PMC - PubMed
-
- Amrein K, Amrein S, Drexler C, Dimai HP, Dobnig H, Pfeifer K, Tomaschitz A, Pieber TR, Fahrleitner-Pammer A. (2012). Sclerostin and its association with physical activity, age, gender, body composition, and bone mineral content in healthy adults. J Clin Endocrinol Metab 97:148-154. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

