PICK1/calcineurin suppress ASIC1-mediated Ca2+ entry in rat pulmonary arterial smooth muscle cells
- PMID: 26702130
- PMCID: PMC4971828
- DOI: 10.1152/ajpcell.00091.2015
PICK1/calcineurin suppress ASIC1-mediated Ca2+ entry in rat pulmonary arterial smooth muscle cells
Abstract
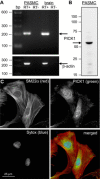
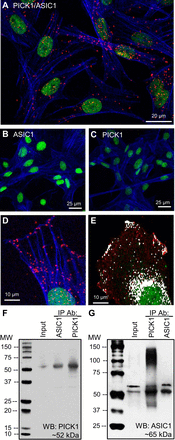
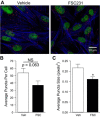
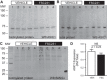
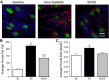
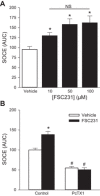
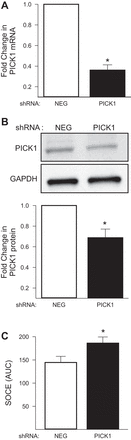
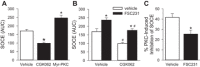
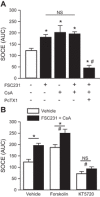
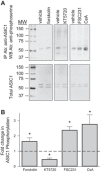
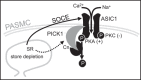
Acid-sensing ion channel 1 (ASIC1) contributes to Ca(2+) influx and contraction in pulmonary arterial smooth muscle cells (PASMC). ASIC1 binds the PDZ (PSD-95/Dlg/ZO-1) domain of the protein interacting with C kinase 1 (PICK1), and this interaction is important for the subcellular localization and/or activity of ASIC1. Therefore, we first hypothesized that PICK1 facilitates ASIC1-dependent Ca(2+) influx in PASMC by promoting plasma membrane localization. Using Duolink to determine protein-protein interactions and a biotinylation assay to assess membrane localization, we demonstrated that the PICK1 PDZ domain inhibitor FSC231 diminished the colocalization of PICK1 and ASIC1 but did not limit ASIC1 plasma membrane localization. Although stimulation of store-operated Ca(2+) entry (SOCE) greatly enhanced colocalization between ASIC1 and PICK1, both FSC231 and shRNA knockdown of PICK1 largely augmented SOCE. These data suggest PICK1 imparts a basal inhibitory effect on ASIC1 Ca(2+) entry in PASMC and led to an alternative hypothesis that PICK1 facilitates the interaction between ASIC1 and negative intracellular modulators, namely PKC and/or the calcium-calmodulin-activated phosphatase calcineurin. FSC231 limited PKC-mediated inhibition of SOCE, supporting a potential role for PICK1 in this response. Additionally, we found PICK1 inhibits ASIC1-mediated SOCE through an effect of calcineurin to dephosphorylate the channel. Furthermore, it appears PICK1/calcineurin-mediated regulation of SOCE opposes PKA phosphorylation and activation of ASIC1. Together our data suggest PKA and PICK1/calcineurin differentially regulate ASIC1-mediated SOCE and these modulatory complexes are important in determining downstream Ca(2+) signaling.
Keywords: DEG/ENaC; Duolink; FSC231; PKA; PKC; store-operated calcium entry.
Copyright © 2016 the American Physiological Society.
Figures
References
-
- Anzai N, Deval E, Schaefer L, Friend V, Lazdunski M, Lingueglia E. The multivalent PDZ domain-containing protein CIPP is a partner of acid-sensing ion channel 3 in sensory neurons. J Biol Chem 277: 16655–16661, 2002. - PubMed
-
- Baron A, Deval E, Salinas M, Lingueglia E, Voilley N, Lazdunski M. Protein kinase C stimulates the acid-sensing ion channel ASIC2a via the PDZ domain-containing protein PICK1. J Biol Chem 277: 50463–50468, 2002. - PubMed
-
- Berdiev BK, Xia J, Jovov B, Markert JM, Mapstone TB, Gillespie GY, Fuller CM, Bubien JK, Benos DJ. Protein kinase C isoform antagonism controls BNaC2 (ASIC1) function. J Biol Chem 277: 45734–45740, 2002. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

