Structure of Guanylyl Cyclase Activator Protein 1 (GCAP1) Mutant V77E in a Ca2+-free/Mg2+-bound Activator State
- PMID: 26703466
- PMCID: PMC4813471
- DOI: 10.1074/jbc.M115.696161
Structure of Guanylyl Cyclase Activator Protein 1 (GCAP1) Mutant V77E in a Ca2+-free/Mg2+-bound Activator State
Abstract
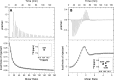
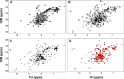
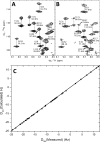
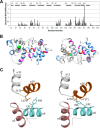
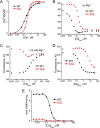
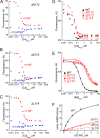
GCAP1, a member of the neuronal calcium sensor subclass of the calmodulin superfamily, confers Ca(2+)-sensitive activation of retinal guanylyl cyclase 1 (RetGC1). We present NMR resonance assignments, residual dipolar coupling data, functional analysis, and a structural model of GCAP1 mutant (GCAP1(V77E)) in the Ca(2+)-free/Mg(2+)-bound state. NMR chemical shifts and residual dipolar coupling data reveal Ca(2+)-dependent differences for residues 170-174. An NMR-derived model of GCAP1(V77E) contains Mg(2+) bound at EF2 and looks similar to Ca(2+) saturated GCAP1 (root mean square deviations = 2.0 Å). Ca(2+)-dependent structural differences occur in the fourth EF-hand (EF4) and adjacent helical region (residues 164-174 called the Ca(2+) switch helix). Ca(2+)-induced shortening of the Ca(2+) switch helix changes solvent accessibility of Thr-171 and Leu-174 that affects the domain interface. Although the Ca(2+) switch helix is not part of the RetGC1 binding site, insertion of an extra Gly residue between Ser-173 and Leu-174 as well as deletion of Arg-172, Ser-173, or Leu-174 all caused a decrease in Ca(2+) binding affinity and abolished RetGC1 activation. We conclude that Ca(2+)-dependent conformational changes in the Ca(2+) switch helix are important for activating RetGC1 and provide further support for a Ca(2+)-myristoyl tug mechanism.
Keywords: calcium; calcium-binding protein; calorimetry; guanylate cyclase (guanylyl cyclase); nuclear magnetic resonance (NMR); phototransduction; vision.
© 2016 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures


References
-
- Ames J. B., Tanaka T., Stryer L., and Ikura M. (1996) Portrait of a myristoyl switch protein. Curr. Opin. Struct. Biol. 6, 432–438 - PubMed
-
- Dizhoor A. M., Lowe D. G., Olshevskaya E. V., Laura R. P., and Hurley J. B. (1994) The human photoreceptor membrane guanylyl cyclase, RetGC, is present in outer segments and is regulated by calcium and a soluble activator. Neuron 12, 1345–1352 - PubMed
-
- Koch K. W., and Stryer L. (1988) Highly cooperative feedback control of retinal rod guanylate cyclase by calcium ions. Nature 334, 64–66 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

