Roles of nucleic acid substrates and cofactors in the vhs protein activity of pseudorabies virus
- PMID: 26704628
- PMCID: PMC4699370
- DOI: 10.1186/s13567-015-0284-y
Roles of nucleic acid substrates and cofactors in the vhs protein activity of pseudorabies virus
Abstract
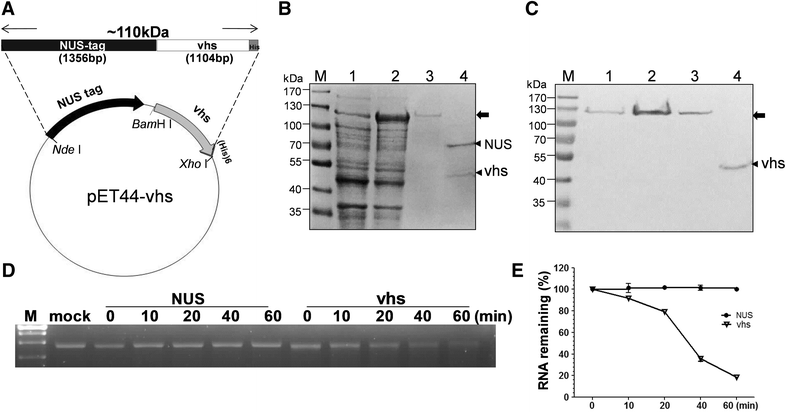
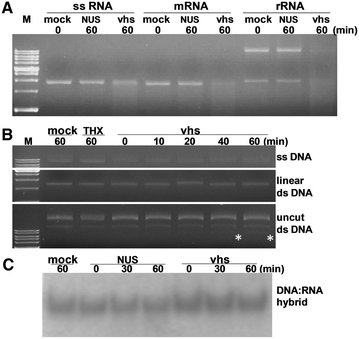
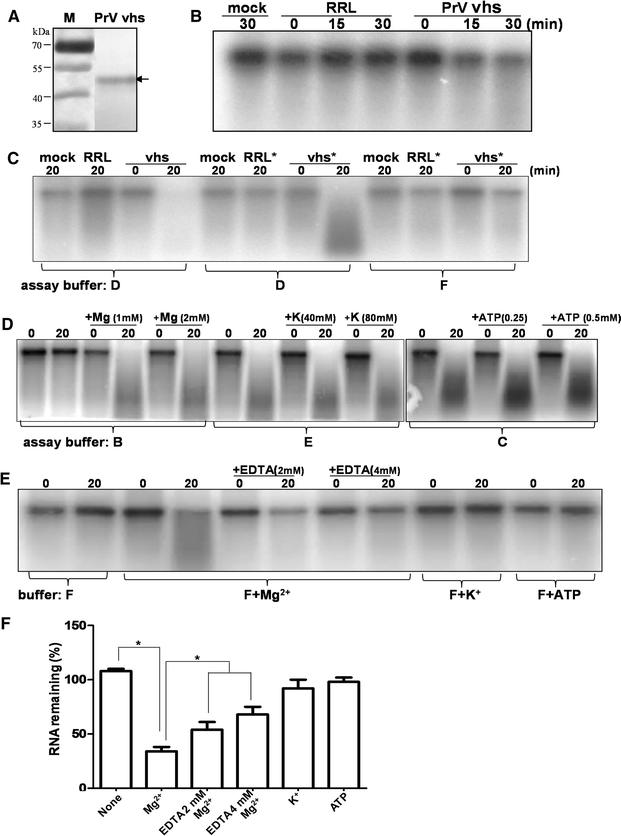
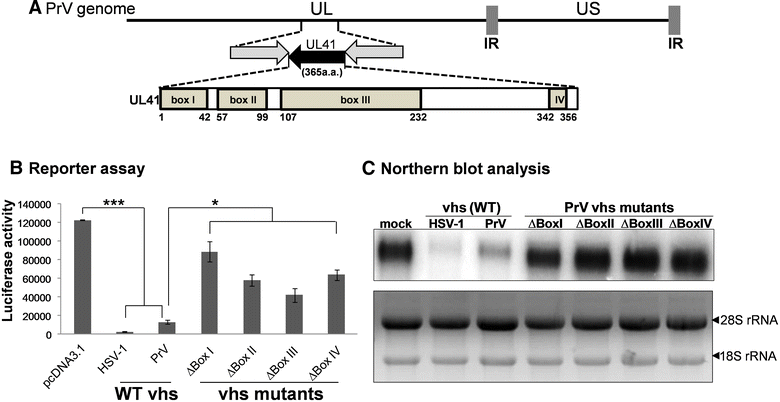
Pseudorabies virus (PrV) belongs to the α-herpesvirinae of which human simplex virus (HSV) is the prototype virus. One of the hallmarks of HSV infection is shutoff of protein synthesis that is mediated by various viral proteins including vhs (virion host shutoff), which is encoded by the UL41 gene. However, the function of PrV vhs is poorly understood. Due to the low sequence similarity (39.3%) between the HSV and PrV UL41 proteins, vhs might not share the same biochemistry characteristics. The purpose of this study was to characterize the nuclease activity of the PrV vhs protein with respect to substrate specificity, its requirements in terms of cofactors, and the protein regions, as well as key amino acids, which contribute to vhs activity. Our results indicated that, similar to HSV vhs, PrV vhs is able to degrade ssRNA and mRNA. However, PrV vhs also targeted rRNA for degradation, which is novel compared to the HSV-1 vhs. Activity assays indicated that Mg(2+) alone enhances RNA degradation mediated by PrV vhs, while K(+) and ATP are not sufficient to induce activity. Finally, we demonstrated that each of the four highly conserved functional boxes of PrV vhs contributes to RNA degradation and that, in particular, residues 152, 169, 171, 172, 173 343, 345, 352 and 356, which are conserved among α-herpesviruses, are key amino acids needed for PrV vhs ribonuclease activity.
Figures

Similar articles
-
The pseudorabies virus vhs protein cleaves RNA containing an IRES sequence.FEBS J. 2016 Mar;283(5):899-911. doi: 10.1111/febs.13642. Epub 2016 Feb 3. FEBS J. 2016. PMID: 26744129
-
Generation and characterization of UL41 null pseudorabies virus variant in vitro and in vivo.Virol J. 2018 Aug 2;15(1):119. doi: 10.1186/s12985-018-1025-4. Virol J. 2018. PMID: 30071879 Free PMC article.
-
Role of the UL41 protein of pseudorabies virus in host shutoff, pathogenesis and induction of TNF-α expression.J Vet Med Sci. 2010 Sep;72(9):1179-87. doi: 10.1292/jvms.10-0059. Epub 2010 Apr 24. J Vet Med Sci. 2010. PMID: 20448414
-
Early shutoff of host protein synthesis in cells infected with herpes simplex viruses.Acta Virol. 2001;45(5-6):269-77. Acta Virol. 2001. PMID: 12083325 Review.
-
Gene and cancer therapy--pseudorabies virus: a novel research and therapeutic tool?Curr Gene Ther. 2003 Apr;3(2):155-82. doi: 10.2174/1566523034578393. Curr Gene Ther. 2003. PMID: 12653408 Review.
Cited by
-
Comprehensive classification of the PIN domain-like superfamily.Nucleic Acids Res. 2017 Jul 7;45(12):6995-7020. doi: 10.1093/nar/gkx494. Nucleic Acids Res. 2017. PMID: 28575517 Free PMC article.
-
Pseudorabies Virus: From Pathogenesis to Prevention Strategies.Viruses. 2022 Jul 27;14(8):1638. doi: 10.3390/v14081638. Viruses. 2022. PMID: 36016260 Free PMC article. Review.
-
IFITM1 is a host restriction factor that inhibits porcine epidemic diarrhea virus infection.J Nanobiotechnology. 2024 Nov 5;22(1):677. doi: 10.1186/s12951-024-02884-9. J Nanobiotechnology. 2024. PMID: 39501328 Free PMC article.
-
Pseudorabies virus VHS protein abrogates interferon responses by blocking NF-κB and IRF3 nuclear translocation.Virol Sin. 2024 Aug;39(4):587-599. doi: 10.1016/j.virs.2024.05.009. Epub 2024 May 30. Virol Sin. 2024. PMID: 38823782 Free PMC article.
References
-
- Mettenleiter TC. Pseudorabies (Aujeszky’s disease) virus: state of the art. August 1993. Acta Vet Hung. 2004;42:153–177. - PubMed
-
- Tigges MA, Leng S, Johnson DC, Burke RL. Human herpes simplex virus (HSV)-specific CD8+ CTL clones recognize HSV-2-infected fibroblasts after treatment with IFN-gamma or when virion host shutoff functions are disabled. J Immunol. 1996;156:3901–3910. - PubMed
-
- Suzutani T, Nagamine M, Shibaki T, Ogasawara M, Yoshida I, Daikoku T, Nishiyama Y, Azuma M. The role of the UL41 gene of herpes simplex virus type 1 in evasion of non-specific host defence mechanisms during primary infection. J Gen Virol. 2000;81:1763–1771. doi: 10.1099/0022-1317-81-7-1763. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

