Effects of tRNA modification on translational accuracy depend on intrinsic codon-anticodon strength
- PMID: 26704976
- PMCID: PMC4770228
- DOI: 10.1093/nar/gkv1506
Effects of tRNA modification on translational accuracy depend on intrinsic codon-anticodon strength
Abstract
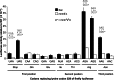
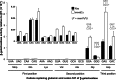
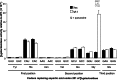
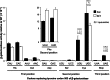
Cellular health and growth requires protein synthesis to be both efficient to ensure sufficient production, and accurate to avoid producing defective or unstable proteins. The background of misreading error frequency by individual tRNAs is as low as 2 × 10(-6) per codon but is codon-specific with some error frequencies above 10(-3) per codon. Here we test the effect on error frequency of blocking post-transcriptional modifications of the anticodon loops of four tRNAs in Escherichia coli. We find two types of responses to removing modification. Blocking modification of tRNA(UUC)(Glu) and tRNA(QUC)(Asp) increases errors, suggesting that the modifications act at least in part to maintain accuracy. Blocking even identical modifications of tRNA(UUU)(Lys) and tRNA(QUA)(Tyr) has the opposite effect of decreasing errors. One explanation could be that the modifications play opposite roles in modulating misreading by the two classes of tRNAs. Given available evidence that modifications help preorder the anticodon to allow it to recognize the codons, however, the simpler explanation is that unmodified 'weak' tRNAs decode too inefficiently to compete against cognate tRNAs that normally decode target codons, which would reduce the frequency of misreading.
© The Author(s) 2015. Published by Oxford University Press on behalf of Nucleic Acids Research.
Figures
Similar articles
-
Codon-specific effects of tRNA anticodon loop modifications on translational misreading errors in the yeast Saccharomyces cerevisiae.Nucleic Acids Res. 2018 Nov 2;46(19):10331-10339. doi: 10.1093/nar/gky664. Nucleic Acids Res. 2018. PMID: 30060218 Free PMC article.
-
The Importance of Being Modified: The Role of RNA Modifications in Translational Fidelity.Enzymes. 2017;41:1-50. doi: 10.1016/bs.enz.2017.03.005. Epub 2017 Apr 22. Enzymes. 2017. PMID: 28601219 Free PMC article. Review.
-
Posttranscriptional modification to the core of tRNAs modulates translational misreading errors.RNA. 2023 Dec 18;30(1):37-51. doi: 10.1261/rna.079797.123. RNA. 2023. PMID: 37907335 Free PMC article.
-
Molecular Coping Mechanisms: Reprogramming tRNAs To Regulate Codon-Biased Translation of Stress Response Proteins.Acc Chem Res. 2023 Dec 5;56(23):3504-3514. doi: 10.1021/acs.accounts.3c00572. Epub 2023 Nov 22. Acc Chem Res. 2023. PMID: 37992267 Free PMC article.
-
tRNA residues that have coevolved with their anticodon to ensure uniform and accurate codon recognition.Biochimie. 2006 Aug;88(8):943-50. doi: 10.1016/j.biochi.2006.06.005. Epub 2006 Jun 23. Biochimie. 2006. PMID: 16828219 Review.
Cited by
-
An integrated, structure- and energy-based view of the genetic code.Nucleic Acids Res. 2016 Sep 30;44(17):8020-40. doi: 10.1093/nar/gkw608. Epub 2016 Jul 22. Nucleic Acids Res. 2016. PMID: 27448410 Free PMC article. Review.
-
Chemical and Conformational Diversity of Modified Nucleosides Affects tRNA Structure and Function.Biomolecules. 2017 Mar 16;7(1):29. doi: 10.3390/biom7010029. Biomolecules. 2017. PMID: 28300792 Free PMC article. Review.
-
Mapping Post-Transcriptional Modifications onto Transfer Ribonucleic Acid Sequences by Liquid Chromatography Tandem Mass Spectrometry.Biomolecules. 2017 Feb 22;7(1):21. doi: 10.3390/biom7010021. Biomolecules. 2017. PMID: 28241457 Free PMC article. Review.
-
Engineering tRNAs for the Ribosomal Translation of Non-proteinogenic Monomers.Chem Rev. 2024 May 22;124(10):6444-6500. doi: 10.1021/acs.chemrev.3c00894. Epub 2024 Apr 30. Chem Rev. 2024. PMID: 38688034 Free PMC article. Review.
-
Hypoxia induces alterations in tRNA modifications involved in translational control.BMC Biol. 2023 Feb 21;21(1):39. doi: 10.1186/s12915-023-01537-x. BMC Biol. 2023. PMID: 36803965 Free PMC article.
References
-
- Yadavalli S.S., Ibba M. Quality control in aminoacyl-tRNA synthesis its role in translational fidelity. Adv. Protein Chem. Struct. Biol. 2012;86:1–43. - PubMed
-
- Kurland C., Hughes D., Ehrenberg M. Limitations of translational accuracy. In: Neidhardt FC, Curtiss R III, Ingraham JL, Lin ECC, Low KB Jr, Magasanik B, Reznikoff WS, Riley M, Schaechter M, Umbarger HE, editors. Escherichia coli and Salmonella: Cellular and Molecular Biology. Vol. 1. Washington, DC: ASM Press; 1996. pp. 979–1004.
-
- Ogle J.M., Carter A.P., Ramakrishnan V. Insights into the decoding mechanism from recent ribosome structures. Trends Biochem. Sci. 2003;28:259–266. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

