Nitro-fatty acids in cardiovascular regulation and diseases: characteristics and molecular mechanisms
- PMID: 26709810
- PMCID: PMC4748372
- DOI: 10.2741/4425
Nitro-fatty acids in cardiovascular regulation and diseases: characteristics and molecular mechanisms
Abstract
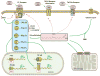
Electrophilic nitro-fatty acids (NO2-FAs) are endogenously formed by redox reactions of nitric oxide ((.)NO)- and nitrite ((.)NO2)- derived nitrogen dioxide with unsaturated fatty acids. Nitration preferentially occurs on polyunsaturated fatty acids with conjugated dienes under physiological or pathophysiological conditions such as during digestion, metabolism and as adaptive inflammatory processes. Nitro-fatty acids are present in free and esterified forms achieving broad biodistribution in humans and experimental models. Structural, functional and biological characterization of NO2-FAs has revealed clinically relevant protection from inflammatory injury in a number of cardiovascular, renal and metabolic experimental models. NO2-FAs are engaged in posttranslational modifications (PTMs) of a selective redox sensitive pool of proteins and regulate key adaptive signaling pathways involved in cellular homeostasis and inflammatory response. Here, we review and update the biosynthesis, metabolism and signaling actions of NO2-FAs, highlighting their diverse protective roles relevant to the cardiovascular system.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

