Solubilization Behavior of Polyene Antibiotics in Nanomicellar System: Insights from Molecular Dynamics Simulation of the Amphotericin B and Nystatin Interactions with Polysorbate 80
- PMID: 26712721
- PMCID: PMC6273564
- DOI: 10.3390/molecules21010006
Solubilization Behavior of Polyene Antibiotics in Nanomicellar System: Insights from Molecular Dynamics Simulation of the Amphotericin B and Nystatin Interactions with Polysorbate 80
Abstract
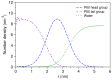
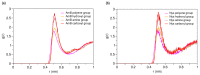
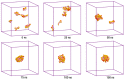
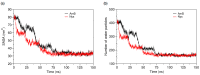
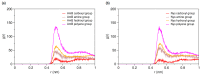
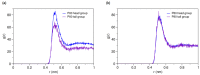
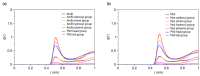
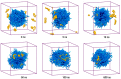
Amphotericin B (AmB) and Nystatin (Nys) are the drugs of choice for treatment of systemic and superficial mycotic infections, respectively, with their full clinical potential unrealized due to the lack of high therapeutic index formulations for their solubilized delivery. In the present study, using a coarse-grained (CG) molecular dynamics (MD) simulation approach, we investigated the interaction of AmB and Nys with Polysorbate 80 (P80) to gain insight into the behavior of these polyene antibiotics (PAs) in nanomicellar solution and derive potential implications for their formulation development. While the encapsulation process was predominantly governed by hydrophobic forces, the dynamics, hydration, localization, orientation, and solvation of PAs in the micelle were largely controlled by hydrophilic interactions. Simulation results rationalized the experimentally observed capability of P80 in solubilizing PAs by indicating (i) the dominant kinetics of drugs encapsulation over self-association; (ii) significantly lower hydration of the drugs at encapsulated state compared with aggregated state; (iii) monomeric solubilization of the drugs; (iv) contribution of drug-micelle interactions to the solubilization; (v) suppressed diffusivity of the encapsulated drugs; (vi) high loading capacity of the micelle; and (vii) the structural robustness of the micelle against drug loading. Supported from the experimental data, our simulations determined the preferred location of PAs to be the core-shell interface at the relatively shallow depth of 75% of micelle radius. Deeper penetration of PAs was impeded by the synergistic effects of (i) limited diffusion of water; and (ii) perpendicular orientation of these drug molecules with respect to the micelle radius. PAs were solvated almost exclusively in the aqueous poly-oxyethylene (POE) medium due to the distance-related lack of interaction with the core, explaining the documented insensitivity of Nys solubilization to drug-core compatibility in detergent micelles. Based on the obtained results, the dearth of water at interior sites of micelle and the large lateral occupation space of PAs lead to shallow insertion, broad radial distribution, and lack of core interactions of the amphiphilic drugs. Hence, controlled promotion of micelle permeability and optimization of chain crowding in palisade layer may help to achieve more efficient solubilization of the PAs.
Keywords: Amphotericin B; Nystatin; Polysorbate 80; drug delivery systems; drug formulation; molecular dynamics simulation; nanomedicine; polyene antibiotics; solubilization.
Conflict of interest statement
The authors declare no conflict of interests.
Figures


Similar articles
-
Insights into specificity and catalytic mechanism of amphotericin B/nystatin thioesterase.Proteins. 2021 May;89(5):558-568. doi: 10.1002/prot.26041. Epub 2021 Jan 15. Proteins. 2021. PMID: 33389775
-
Polysorbate 80 and Cremophor EL micelles deaggregate and solubilize nystatin at the core-corona interface.J Pharm Sci. 2005 Nov;94(11):2345-54. doi: 10.1002/jps.20301. J Pharm Sci. 2005. PMID: 16200548
-
Coarse-Grained Molecular Dynamics Simulation of Heterogeneous Polysorbate 80 Surfactants and their Interactions with Small Molecules and Proteins.Mol Pharm. 2024 Oct 7;21(10):5041-5052. doi: 10.1021/acs.molpharmaceut.4c00461. Epub 2024 Aug 29. Mol Pharm. 2024. PMID: 39208298
-
Pharmaceutical design of the liposomal antimicrobial agents for infectious disease.Curr Pharm Des. 2002;8(6):441-54. doi: 10.2174/1381612023395853. Curr Pharm Des. 2002. PMID: 12069381 Review.
-
Polymeric micelles for delivery of poorly water-soluble compounds.Crit Rev Ther Drug Carrier Syst. 2003;20(5):357-403. doi: 10.1615/critrevtherdrugcarriersyst.v20.i5.20. Crit Rev Ther Drug Carrier Syst. 2003. PMID: 14959789 Review.
Cited by
-
EDTA aggregates induce SYPRO orange-based fluorescence in thermal shift assay.PLoS One. 2017 May 4;12(5):e0177024. doi: 10.1371/journal.pone.0177024. eCollection 2017. PLoS One. 2017. PMID: 28472107 Free PMC article.
-
Intrinsic lipolysis rate for systematic design of lipid-based formulations.Drug Deliv Transl Res. 2023 May;13(5):1288-1304. doi: 10.1007/s13346-022-01246-y. Epub 2022 Oct 8. Drug Deliv Transl Res. 2023. PMID: 36209313 Free PMC article.
-
The Efficacy of Nigella sativa L. and Curcumin Nanomicelle Alone or Together on Lipid Profile, Glycemic Control Indices, and Serum 17-Β Estradiol in Postmenopausal Women.J Caring Sci. 2023 Aug 6;12(3):163-173. doi: 10.34172/jcs.2023.31875. eCollection 2023 Sep. J Caring Sci. 2023. PMID: 38020740 Free PMC article.
-
Novel nanomicelle formulation to enhance bioavailability and stability of curcuminoids.Iran J Basic Med Sci. 2019 Mar;22(3):282-289. doi: 10.22038/ijbms.2019.32873.7852. Iran J Basic Med Sci. 2019. PMID: 31156789 Free PMC article.
-
Self-assembled nanostructures of L-ascorbic acid alkyl esters support monomeric amphotericin B.Heliyon. 2021 Jan 28;7(1):e06056. doi: 10.1016/j.heliyon.2021.e06056. eCollection 2021 Jan. Heliyon. 2021. PMID: 33553743 Free PMC article.
References
-
- Kagan S., Ickowicz D., Shmuel M., Altschuler Y., Sionov E., Pitusi M., Weiss A., Farber S., Domb A.J., Polacheck I. Toxicity mechanisms of amphotericin B and its neutralization by conjugation with arabinogalactan. Antimicrob. Agents Chemother. 2012;56:5603–5611. doi: 10.1128/AAC.00612-12. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

