Roles of Asp179 and Glu270 in ADP-Ribosylation of Actin by Clostridium perfringens Iota Toxin
- PMID: 26713879
- PMCID: PMC4699905
- DOI: 10.1371/journal.pone.0145708
Roles of Asp179 and Glu270 in ADP-Ribosylation of Actin by Clostridium perfringens Iota Toxin
Abstract
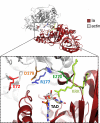
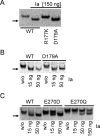
Clostridium perfringens iota toxin is a binary toxin composed of the enzymatically active component Ia and receptor binding component Ib. Ia is an ADP-ribosyltransferase, which modifies Arg177 of actin. The previously determined crystal structure of the actin-Ia complex suggested involvement of Asp179 of actin in the ADP-ribosylation reaction. To gain more insights into the structural requirements of actin to serve as a substrate for toxin-catalyzed ADP-ribosylation, we engineered Saccharomyces cerevisiae strains, in which wild type actin was replaced by actin variants with substitutions in residues located on the Ia-actin interface. Expression of the actin mutant Arg177Lys resulted in complete resistance towards Ia. Actin mutation of Asp179 did not change Ia-induced ADP-ribosylation and growth inhibition of S. cerevisiae. By contrast, substitution of Glu270 of actin inhibited the toxic action of Ia and the ADP-ribosylation of actin. In vitro transcribed/translated human β-actin confirmed the crucial role of Glu270 in ADP-ribosylation of actin by Ia.
Conflict of interest statement
Figures



References
-
- Marvaud JC, Stiles BG, Chenal A, Gillet D, Gibert M, Smith LA, et al. Clostridium perfringens iota toxin. Mapping of the Ia domain involved in docking with Ib and cellular internalization. J Biol Chem. 2002;277:43659–43666. - PubMed
-
- Barth H, Blöcker D, Behlke J, Bergsma-Schutter W, Brisson A, Benz R, et al. Cellular uptake of Clostridium botulinum C2 toxin requires oligomerization and acidification. J Biol Chem. 2000;275:18704–18711. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

