A proposed route to independent measurements of tight junction conductance at discrete cell junctions
- PMID: 26716077
- PMCID: PMC4681292
- DOI: 10.1080/21688370.2015.1105907
A proposed route to independent measurements of tight junction conductance at discrete cell junctions
Abstract
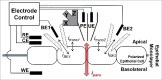
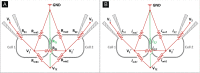
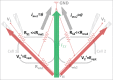
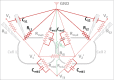
Direct recording of tight junction permeability is of pivotal importance to many biologic fields. Previous approaches bear an intrinsic disadvantage due to the difficulty of separating tight junction conductance from nearby membrane conductance. Here, we propose the design of Double whole-cell Voltage Clamp - Ion Conductance Microscopy (DVC-ICM) based on previously demonstrated potentiometric scanning of local conductive pathways. As proposed, DVC-ICM utilizes two coordinated whole-cell patch-clamps to neutralize the apical membrane current during potentiometric scanning, which in models described here will profoundly enhance the specificity of tight junction recording. Several potential pitfalls are considered, evaluated and addressed with alternative countermeasures.
Keywords: SICM; conductance; ion channel; patch clamp; tight junction.
Figures

References
-
- Farquhar MG, Palade GE. Junctional complexes in various epithelia. J Cell Biol 1963; 17, 375-412; http://dx.doi.org/ 10.1083/jcb.17.2.375 - DOI - PMC - PubMed
-
- Goodenough DA, Revel JP. A. Fine structural analysis of intercellular junctions in the mouse liver. J Cell Biol 1970; 45:272-90; PMID:4105112; http://dx.doi.org/ 10.1083/jcb.45.2.272 - DOI - PMC - PubMed
-
- Furuse M, Hirase T, Itoh M, Nagafuchi A, Yonemura S, Tsukita S, Tsukita S. Occludin: a novel integral membrane protein localizing at tight junctions. J Cell Biol 1993; 123:1777-88; PMID:8276896; http://dx.doi.org/ 10.1083/jcb.123.6.1777 - DOI - PMC - PubMed
-
- Ebnet K, Suzuki A, Ohno S, Vestweber D. Junctional adhesion molecules (JAMs): more molecules with dual functions? J Cell Sci 2004; 117:19-29; PMID:14657270; http://dx.doi.org/ 10.1242/jcs.00930 - DOI - PubMed
-
- Furuse M, Fujita K, Hiiragi T, Fujimoto K, TsuKita S. Claudin-1 and-2: novel integral membrane proteins localizing at tight junctions with no sequence similarity to occludin. J Cell Biol 1998; 141:1539-50; PMID:9647647; http://dx.doi.org/ 10.1083/jcb.141.7.1539 - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
