A bifunctional spin label reports the structural topology of phospholamban in magnetically-aligned bicelles
- PMID: 26720587
- PMCID: PMC4716873
- DOI: 10.1016/j.jmr.2015.12.005
A bifunctional spin label reports the structural topology of phospholamban in magnetically-aligned bicelles
Abstract
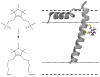
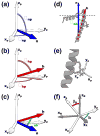
We have applied a bifunctional spin label and EPR spectroscopy to determine membrane protein structural topology in magnetically-aligned bicelles, using monomeric phospholamban (PLB) as a model system. Bicelles are a powerful tool for studying membrane proteins by NMR and EPR spectroscopies, where magnetic alignment yields topological constraints by resolving the anisotropic spectral properties of nuclear and electron spins. However, EPR bicelle studies are often hindered by the rotational mobility of monofunctional Cys-linked spin labels, which obscures their orientation relative to the protein backbone. The rigid and stereospecific TOAC label provides high orientational sensitivity but must be introduced via solid-phase peptide synthesis, precluding its use in large proteins. Here we show that a bifunctional methanethiosulfonate spin label attaches rigidly and stereospecifically to Cys residues at i and i+4 positions along PLB's transmembrane helix, thus providing orientational resolution similar to that of TOAC, while being applicable to larger membrane proteins for which synthesis is impractical. Computational modeling and comparison with NMR data shows that these EPR experiments provide accurate information about helix tilt relative to the membrane normal, thus establishing a robust method for determining structural topology in large membrane proteins with a substantial advantage in sensitivity over NMR.
Keywords: Bicelles; Bifunctional spin label; EPR; Molecular dynamics; Orientation; Phospholamban.
Copyright © 2015 Elsevier Inc. All rights reserved.
Figures
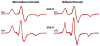
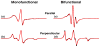
References
-
- Traaseth NJ, et al. Structural dynamics and topology of phospholamban in oriented lipid bilayers using multidimensional solid-state NMR. Biochemistry. 2006;45(46):13827–34. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

