In vivo genome editing improves muscle function in a mouse model of Duchenne muscular dystrophy
- PMID: 26721684
- PMCID: PMC4883596
- DOI: 10.1126/science.aad5143
In vivo genome editing improves muscle function in a mouse model of Duchenne muscular dystrophy
Abstract
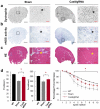
Duchenne muscular dystrophy (DMD) is a devastating disease affecting about 1 out of 5000 male births and caused by mutations in the dystrophin gene. Genome editing has the potential to restore expression of a modified dystrophin gene from the native locus to modulate disease progression. In this study, adeno-associated virus was used to deliver the clustered regularly interspaced short palindromic repeats (CRISPR)-Cas9 system to the mdx mouse model of DMD to remove the mutated exon 23 from the dystrophin gene. This includes local and systemic delivery to adult mice and systemic delivery to neonatal mice. Exon 23 deletion by CRISPR-Cas9 resulted in expression of the modified dystrophin gene, partial recovery of functional dystrophin protein in skeletal myofibers and cardiac muscle, improvement of muscle biochemistry, and significant enhancement of muscle force. This work establishes CRISPR-Cas9-based genome editing as a potential therapy to treat DMD.
Copyright © 2016, American Association for the Advancement of Science.
Figures



Comment in
-
Cautious welcome for gene editing of Duchenne muscular dystrophy in animal model.BMJ. 2016 Jan 4;351:h7033. doi: 10.1136/bmj.h7033. BMJ. 2016. PMID: 26729900 No abstract available.
-
Genetic engineering: In vivo genome editing - growing in strength.Nat Rev Genet. 2016 Mar;17(3):124. doi: 10.1038/nrg.2016.2. Epub 2016 Jan 19. Nat Rev Genet. 2016. PMID: 26781811 No abstract available.
-
Exon Snipping in Duchenne Muscular Dystrophy.Trends Mol Med. 2016 Mar;22(3):187-189. doi: 10.1016/j.molmed.2016.01.007. Epub 2016 Feb 5. Trends Mol Med. 2016. PMID: 26856237
-
CRISPR/Cas9 Flexes Its Muscles: In Vivo Somatic Gene Editing for Muscular Dystrophy.Mol Ther. 2016 Mar;24(3):414-6. doi: 10.1038/mt.2016.29. Mol Ther. 2016. PMID: 26952918 Free PMC article. No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
- R01 NS090634/NS/NINDS NIH HHS/United States
- UL1 TR001111/TR/NCATS NIH HHS/United States
- DP2 OD008586/OD/NIH HHS/United States
- R01 MH110049/MH/NIMH NIH HHS/United States
- DP1-MH100706/DP/NCCDPHP CDC HHS/United States
- T32 GM007753/GM/NIGMS NIH HHS/United States
- UL1 TR002489/TR/NCATS NIH HHS/United States
- P01HL112761/HL/NHLBI NIH HHS/United States
- DP2-OD008586/OD/NIH HHS/United States
- DP1 MH100706/MH/NIMH NIH HHS/United States
- R01NS90634/NS/NINDS NIH HHS/United States
- R01 DK097768/DK/NIDDK NIH HHS/United States
- T32 GM008313/GM/NIGMS NIH HHS/United States
- T32GM007753/GM/NIGMS NIH HHS/United States
- P01 HL112761/HL/NHLBI NIH HHS/United States
- R01HL089221/HL/NHLBI NIH HHS/United States
- R01DK097768/DK/NIDDK NIH HHS/United States
- R01 HL089221/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

