Interleukin-18 modulation in autism spectrum disorders
- PMID: 26728085
- PMCID: PMC4700739
- DOI: 10.1186/s12974-015-0466-6
Interleukin-18 modulation in autism spectrum disorders
Abstract
Background: Autism spectrum disorder (ASD) is a neurodevelopmental disease which affects 1 in 88 children. Its etiology remains basically unknown, but it is apparent that neuroinflammation is involved in disease development. Great attention has been focused on pro-inflammatory cytokines, and several studies have reported their dysfunction unbalance in serum as well as in the brain. The present work aimed at evaluating putative dysregulation of interleukin-18 (IL-18), a pro-inflammatory cytokine of the IL-1 family in the sera of patients with ASD of different grades, compared to healthy controls, as well as in postmortem brain samples obtained from patients with tuberous sclerosis as well as acute inflammatory diseases. Moreover, quantitative analysis of IL-18 was performed in the sera and brain obtained from Reeler mice, an experimental model of autism.
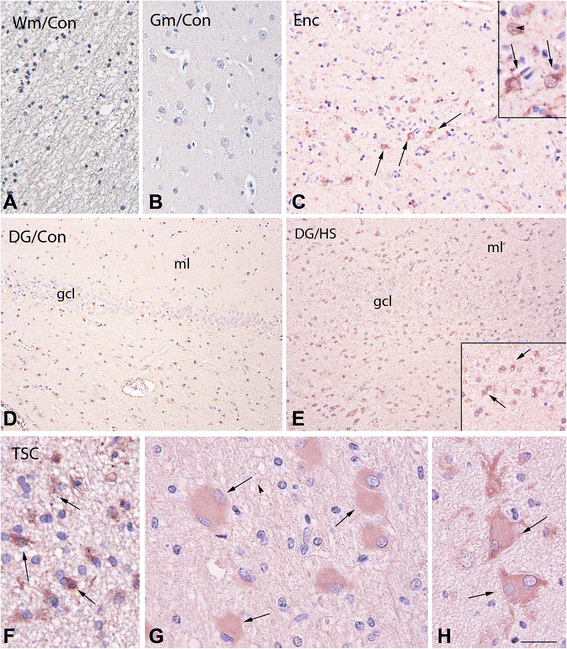
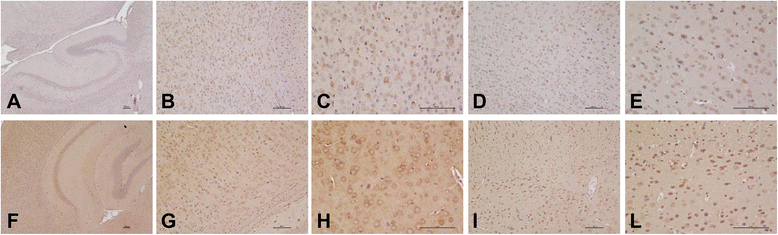
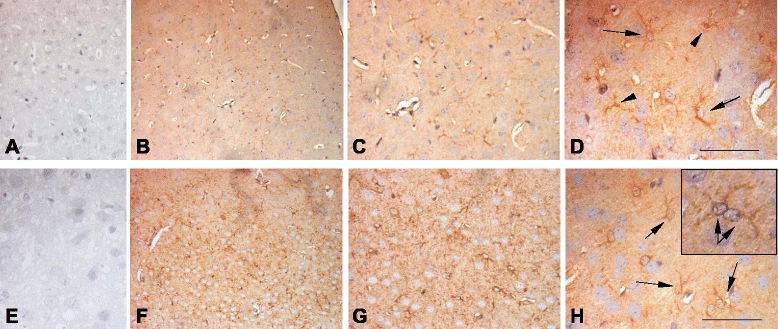
Methods: Serum IL-18 levels were measured by ELISA. IL-18 was localized by immunohistochemical analysis in brain sections obtained from tuberous sclerosis and encephalitis patients, as well as from gender- and age-matched controls, and in the brain sections of both Reeler and wild-type mice. IL-18 was also quantified by Western blots in homogenates of Reeler and wild-type mice brains. IL-18 binding protein (IL-18BP) was evaluated in Reeler and wild-type mice plasma as well as in their brains (sections and homogenates).
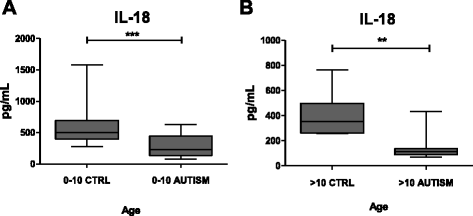
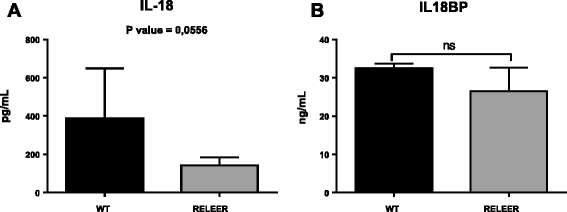
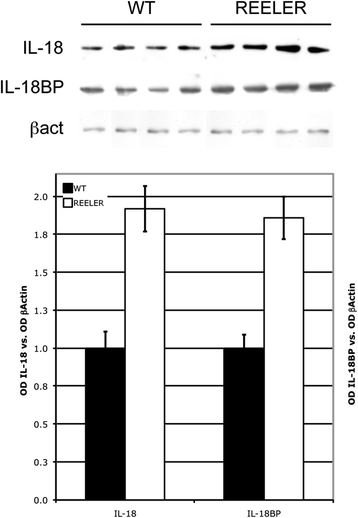
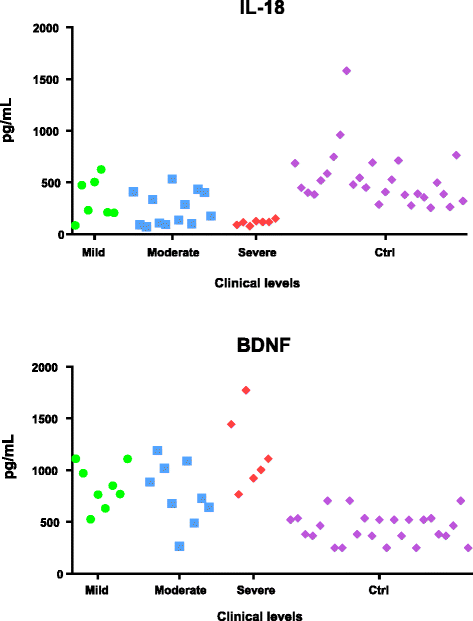
Results: IL-18 content decreased in the sera of patients with autism compared to healthy subjects and in Reeler sera compared to wild-type controls. IL-18 was detected within glial cells and neurons in the brain of subjects affected by tuberous sclerosis and encephalitis whereas in healthy subjects, only a weak IL-18 positivity was detected at the level of glial cells. Western blot identified higher amounts of IL-18 in Reeler brain homogenates compared to wild-type littermates. IL-18BP was expressed in higher amounts in Reeler brain compared to the brain of wild-type mice, whereas no significant difference was detected comparing IL-18BP plasma levels.
Conclusions: IL-18 is dysregulated in ASD patients. Further studies seemed necessary to clarify the molecular details behind IL-18 increase in the brain and IL-18 decrease in the sera of patients. An increase in the size of the patient cohort seems necessary to ascertain whether decreased IL-18 content in the sera can become a predictive biomarker of ASD and whether its measure, in combination with other markers (e.g., increased levels of brain-derived neurotrophic factor (BDNF)), may be included in a diagnostic panel.
Figures
Similar articles
-
Elevated serum levels of free interleukin-18 in adult-onset Still's disease.Rheumatology (Oxford). 2016 Dec;55(12):2237-2247. doi: 10.1093/rheumatology/kew300. Epub 2016 Sep 10. Rheumatology (Oxford). 2016. PMID: 27616144
-
Increased serum levels of brain-derived neurotrophic factor in autism spectrum disorder.Neuroreport. 2015 Aug 5;26(11):638-41. doi: 10.1097/WNR.0000000000000404. Neuroreport. 2015. PMID: 26103118
-
Serum IL-18 and IL-18BP levels in patients with Chikungunya virus infection.Viral Immunol. 2010 Feb;23(1):113-7. doi: 10.1089/vim.2009.0077. Viral Immunol. 2010. PMID: 20121409
-
A meta-analysis of pro-inflammatory cytokines in autism spectrum disorders: Effects of age, gender, and latitude.J Psychiatr Res. 2019 Aug;115:90-102. doi: 10.1016/j.jpsychires.2019.05.019. Epub 2019 May 18. J Psychiatr Res. 2019. PMID: 31125917
-
Neuroimmunologic and Neurotrophic Interactions in Autism Spectrum Disorders: Relationship to Neuroinflammation.Neuromolecular Med. 2018 Jun;20(2):161-173. doi: 10.1007/s12017-018-8488-8. Epub 2018 Apr 24. Neuromolecular Med. 2018. PMID: 29691724 Free PMC article. Review.
Cited by
-
Traffic-related particulate matter affects behavior, inflammation, and neural integrity in a developmental rodent model.Environ Res. 2020 Apr;183:109242. doi: 10.1016/j.envres.2020.109242. Epub 2020 Feb 17. Environ Res. 2020. PMID: 32097814 Free PMC article.
-
Dual Profile of Environmental Enrichment and Autistic-Like Behaviors in the Maternal Separated Model in Rats.Int J Mol Sci. 2021 Jan 25;22(3):1173. doi: 10.3390/ijms22031173. Int J Mol Sci. 2021. PMID: 33503967 Free PMC article.
-
Crosstalk Between Mitochondrial DNA and Immune Response: Focus on Autism Spectrum Disorder.Mol Neurobiol. 2025 May;62(5):5629-5639. doi: 10.1007/s12035-024-04637-z. Epub 2024 Nov 26. Mol Neurobiol. 2025. PMID: 39589631 Review.
-
The Role of the Gut Microbiota and the Immune System in the Development of Autism.Cureus. 2020 Oct 28;12(10):e11226. doi: 10.7759/cureus.11226. Cureus. 2020. PMID: 33269154 Free PMC article. Review.
-
Comprehensive literature data-mining analysis reveals a broad genetic network functionally associated with autism spectrum disorder.Int J Mol Med. 2018 Nov;42(5):2353-2362. doi: 10.3892/ijmm.2018.3845. Epub 2018 Aug 28. Int J Mol Med. 2018. PMID: 30226572 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

