Achievements, challenges and unmet needs for haemophilia patients with inhibitors: Report from a symposium in Paris, France on 20 November 2014
- PMID: 26728503
- PMCID: PMC4989271
- DOI: 10.1111/hae.12860
Achievements, challenges and unmet needs for haemophilia patients with inhibitors: Report from a symposium in Paris, France on 20 November 2014
Abstract
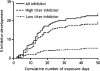
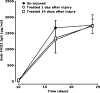
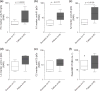
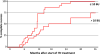
Over the past 20 years, there have been many advances in haemophilia treatment that have allowed patients to take greater control of their disease. However, the development of factor VIII (FVIII) inhibitors is the greatest complication of the disease and a challenge in the treatment of haemophilia making management of bleeding episodes difficult and surgical procedures very challenging. A meeting to discuss the unmet needs of haemophilia patients with inhibitors was held in Paris on 20 November 2014. Topics discussed were genetic and non-genetic risk factors for the development of inhibitors, immunological aspects of inhibitor development, FVIII products and inhibitor development, generation and functional properties of engineered antigen-specific T regulatory cells, suppression of immune responses to FVIII, prophylaxis in haemophilia patients with inhibitors, epitope mapping of FVIII inhibitors, current controversies in immune tolerance induction therapy, surgery in haemophilia patients with inhibitors and future perspectives for the treatment of haemophilia patients with inhibitors. A summary of the key points discussed is presented in this paper.
Keywords: FVIII products; genetics; haemophilia; inhibitors; prophylaxis; surgery.
© 2016 John Wiley & Sons Ltd.
Figures


Similar articles
-
Factor VIII-specific B cell responses in haemophilia A patients with inhibitors.Haemophilia. 2010 May;16(102):35-43. doi: 10.1111/j.1365-2516.2010.02215.x. Haemophilia. 2010. PMID: 20536984 Review.
-
Mechanisms of action of immune tolerance induction against factor VIII in patients with congenital haemophilia A and factor VIII inhibitors.Br J Haematol. 2007 Jan;136(1):12-25. doi: 10.1111/j.1365-2141.2006.06359.x. Br J Haematol. 2007. PMID: 17222196 Review.
-
Neutralisation of factor VIII inhibitors by anti-idiotypes isolated from phage-displayed libraries.Thromb Haemost. 2016 Jul 4;116(1):32-41. doi: 10.1160/TH15-12-0925. Epub 2016 Mar 24. Thromb Haemost. 2016. PMID: 27009573
-
Regulatory T cells and their potential for tolerance induction in haemophilia A patients.Hamostaseologie. 2016 Nov 8;36(Suppl. 2):S5-S12. Hamostaseologie. 2016. PMID: 27824212 Review.
-
Prophylactic bypassing agent use before and during immune tolerance induction in patients with haemophilia A and inhibitors to FVIII.Haemophilia. 2018 Jul;24(4):570-577. doi: 10.1111/hae.13534. Epub 2018 Jun 14. Haemophilia. 2018. PMID: 29901839 Review.
Cited by
-
The New Paradigms in Clinical Research: From Early Access Programs to the Novel Therapeutic Approaches for Unmet Medical Needs.Front Pharmacol. 2019 Feb 13;10:111. doi: 10.3389/fphar.2019.00111. eCollection 2019. Front Pharmacol. 2019. PMID: 30814951 Free PMC article. Review.
-
Impact of the Survey of Inhibitors in Plasma-Product Exposed Toddlers (SIPPET) study and its post hoc analyses on clinical practice in the United States: A survey of Haemophilia and Thrombosis Research Society members.Haemophilia. 2019 Sep;25(5):764-772. doi: 10.1111/hae.13806. Epub 2019 Jul 2. Haemophilia. 2019. PMID: 31264762 Free PMC article.
-
Impacts of the COVID-19 pandemic on pediatric and adult patients with hemophilia.Ann Hematol. 2025 Jan;104(1):95-104. doi: 10.1007/s00277-024-05992-6. Epub 2024 Sep 14. Ann Hematol. 2025. PMID: 39271522 Free PMC article.
-
Induction of activated T follicular helper cells is critical for anti-FVIII inhibitor development in hemophilia A mice.Blood Adv. 2019 Oct 22;3(20):3099-3110. doi: 10.1182/bloodadvances.2019000650. Blood Adv. 2019. PMID: 31648333 Free PMC article.
-
MG1113, a specific anti-tissue factor pathway inhibitor antibody, rebalances the coagulation system and promotes hemostasis in hemophilia.Res Pract Thromb Haemost. 2020 Oct 22;4(8):1301-1312. doi: 10.1002/rth2.12438. eCollection 2020 Nov. Res Pract Thromb Haemost. 2020. PMID: 33313469 Free PMC article.
References
-
- Saint-Remy JM, Lacroix-Desmazes S, Oldenburg J. Inhibitors in haemophilia: pathophysiology. Haemophilia. 2004;10(Suppl 4):146–51. - PubMed
-
- Lozner EL, Joliffe LS, Taylor FHL. Hemorrhagic diathesis with prolonged coagulation time, associated with a circulating anticoagulant. Am J Med Sci. 1940;199:318.
-
- Gitschier J, Wood WI, Tuddenham EG, et al. Detection and sequence of mutations in the factor VIII gene of haemophiliacs. Nature. 1985;315:427–30. - PubMed
-
- Oldenburg J, Picard JK, Schwaab R, Brackmann HH, Tuddenham EG, Simpson E. HLA genotype of patients with severe haemophilia A due to intron 22 inversion with and without inhibitors of factor VIII. Thromb Haemost. 1997;77:238–42. - PubMed
-
- Astermark J, Oldenburg J, Carlson J, et al. Polymorphisms in the TNFA gene and the risk of inhibitor development in patients with hemophilia A. Blood. 2006;108:3739–45. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

