Development of a Vaccine against Escherichia coli Urinary Tract Infections
- PMID: 26729174
- PMCID: PMC4810122
- DOI: 10.3390/pathogens5010001
Development of a Vaccine against Escherichia coli Urinary Tract Infections
Abstract
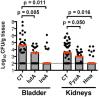
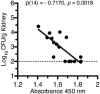
Urinary tract infection (UTI) is the second most common infection in humans after those involving the respiratory tract. This results not only in huge annual economic costs, but in decreased workforce productivity and high patient morbidity. Most infections are caused by uropathogenic Escherichia coli (UPEC). Antibiotic treatment is generally effective for eradication of the infecting strain; however, documentation of increasing antibiotic resistance, allergic reaction to certain pharmaceuticals, alteration of normal gut flora, and failure to prevent recurrent infections represent significant barriers to treatment. As a result, approaches to prevent UTI such as vaccination represent a gap that must be addressed. Our laboratory has made progress toward development of a preventive vaccine against UPEC. The long-term research goal is to prevent UTIs in women with recurrent UTIs. Our objective has been to identify the optimal combination of protective antigens for inclusion in an effective UTI vaccine, optimal adjuvant, optimal dose, and optimal route of delivery. We hypothesized that a multi-subunit vaccine elicits antibody that protects against experimental challenge with UPEC strains. We have systematically identified four antigens that can individually protect experimentally infected mice from colonization of the bladder and/or kidneys by UPEC when administered intranasally with cholera toxin (CT) as an adjuvant. To advance the vaccine for utility in humans, we will group the individual antigens, all associated with iron acquisition (IreA, Hma, IutA, FyuA), into an effective combination to establish a multi-subunit vaccine. We demonstrated for all four vaccine antigens that antigen-specific serum IgG represents a strong correlate of protection in vaccinated mice. High antibody titers correlate with low colony forming units (CFUs) of UPEC following transurethral challenge of vaccinated mice. However, the contribution of cell-mediated immunity cannot be ruled out and must be investigated experimentally. We have demonstrated that antibodies bind to the surface of UPEC expressing the antigens. Sera from women with and without histories of UTI have been tested for antibody levels to vaccine antigens. Our results validate iron acquisition as a target for vaccination against UTI.
Keywords: E. coli; antibody response; urinary tract infection; vaccine.
Figures

References
-
- Schappert S.M., Rechtsteiner E.A. Ambulatory medical care utilization estimates for 2006. [(accessed on 30 December 2015)]. Available online: http://198.246.124.22/nchs/data/nhsr/nhsr008.pdf. - PubMed
-
- O’Hanley P. Prospects for urinary tract infection vaccines. In: Mobley H.L.T., Warren J.W., editors. Urinary Tract Infections: Molecular Pathogenesis and Clinical Management. ASM Press; Washington, DC, USA: 1996. pp. 405–425.
-
- Litwin M.S., Saigal C.S. Introduction. GPO; Washington, DC, USA: 2007.
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

