A Common Genetic Variant in TLR1 Enhances Human Neutrophil Priming and Impacts Length of Intensive Care Stay in Pediatric Sepsis
- PMID: 26729809
- PMCID: PMC4724530
- DOI: 10.4049/jimmunol.1500856
A Common Genetic Variant in TLR1 Enhances Human Neutrophil Priming and Impacts Length of Intensive Care Stay in Pediatric Sepsis
Abstract
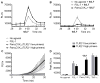
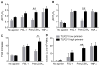
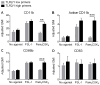
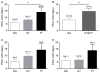
Polymorphonuclear leukocytes (PMN) achieve an intermediate or primed state of activation following stimulation with certain agonists. Primed PMN have enhanced responsiveness to subsequent stimuli, which can be beneficial in eliminating microbes but may cause host tissue damage in certain disease contexts, including sepsis. As PMN priming by TLR4 agonists is well described, we hypothesized that ligation of TLR2/1 or TLR2/6 would prime PMN. Surprisingly, PMN from only a subset of donors were primed in response to the TLR2/1 agonist, Pam3CSK4, although PMN from all donors were primed by the TLR2/6 agonist, FSL-1. Priming responses included generation of intracellular and extracellular reactive oxygen species, MAPK phosphorylation, integrin activation, secondary granule exocytosis, and cytokine secretion. Genotyping studies revealed that PMN responsiveness to Pam3CSK4 was enhanced by a common single-nucleotide polymorphism (SNP) in TLR1 (rs5743618). Notably, PMN from donors with the SNP had higher surface levels of TLR1 and were demonstrated to have enhanced association of TLR1 with the endoplasmic reticulum chaperone gp96. We analyzed TLR1 genotypes in a pediatric sepsis database and found that patients with sepsis or septic shock who had a positive blood culture and were homozygous for the SNP associated with neutrophil priming had prolonged pediatric intensive care unit length of stay. We conclude that this TLR1 SNP leads to excessive PMN priming in response to cell stimulation. Based on our finding that septic children with this SNP had longer pediatric intensive care unit stays, we speculate that this SNP results in hyperinflammation in diseases such as sepsis.
Copyright © 2016 by The American Association of Immunologists, Inc.
Conflict of interest statement
The authors have declared that no conflict of interest exists.
Figures
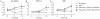
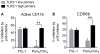
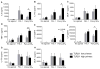
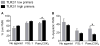
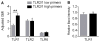
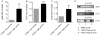
References
-
- Moreland JG, Davis AP, Matsuda JJ, Hook JS, Bailey G, Nauseef WM, Lamb FS. Endotoxin priming of neutrophils requires NADPH oxidase-generated oxidants and is regulated by the anion transporter ClC-3. The Journal of biological chemistry. 2007;282:33958–33967. - PubMed
-
- Weigand MA, Horner C, Bardenheuer HJ, Bouchon A. The systemic inflammatory response syndrome. Best Pract Res Clin Anaesthesiol. 2004;18:455–475. - PubMed
-
- Uciechowski P, Imhoff H, Lange C, Meyer CG, Browne EN, Kirsten DK, Schroder AK, Schaaf B, Al-Lahham A, Reinert RR, Reiling N, Haase H, Hatzmann A, Fleischer D, Heussen N, Kleines M, Rink L. Susceptibility to tuberculosis is associated with TLR1 polymorphisms resulting in a lack of TLR1 cell surface expression. J Leukoc Biol. 2011;90:377–388. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

