Corynebacterium accolens Releases Antipneumococcal Free Fatty Acids from Human Nostril and Skin Surface Triacylglycerols
- PMID: 26733066
- PMCID: PMC4725001
- DOI: 10.1128/mBio.01725-15
Corynebacterium accolens Releases Antipneumococcal Free Fatty Acids from Human Nostril and Skin Surface Triacylglycerols
Abstract
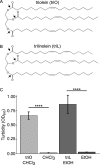
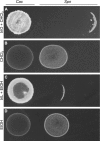
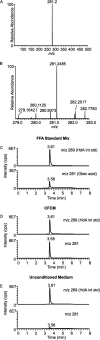
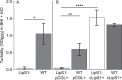
Bacterial interspecies interactions play clinically important roles in shaping microbial community composition. We observed that Corynebacterium spp. are overrepresented in children free of Streptococcus pneumoniae (pneumococcus), a common pediatric nasal colonizer and an important infectious agent. Corynebacterium accolens, a benign lipid-requiring species, inhibits pneumococcal growth during in vitro cocultivation on medium supplemented with human skin surface triacylglycerols (TAGs) that are likely present in the nostrils. This inhibition depends on LipS1, a TAG lipase necessary for C. accolens growth on TAGs such as triolein. We determined that C. accolens hydrolysis of triolein releases oleic acid, which inhibits pneumococcus, as do other free fatty acids (FFAs) that might be released by LipS1 from human skin surface TAGs. Our results support a model in which C. accolens hydrolyzes skin surface TAGS in vivo releasing antipneumococcal FFAs. These data indicate that C. accolens may play a beneficial role in sculpting the human microbiome.
Importance: Little is known about how harmless Corynebacterium species that colonize the human nose and skin might impact pathogen colonization and proliferation at these sites. We show that Corynebacterium accolens, a common benign nasal bacterium, modifies its local habitat in vitro as it inhibits growth of Streptococcus pneumoniae by releasing antibacterial free fatty acids from host skin surface triacylglycerols. We further identify the primary C. accolens lipase required for this activity. We postulate a model in which higher numbers of C. accolens cells deter/limit S. pneumoniae nostril colonization, which might partly explain why children without S. pneumoniae colonization have higher levels of nasal Corynebacterium. This work narrows the gap between descriptive studies and the needed in-depth understanding of the molecular mechanisms of microbe-microbe interactions that help shape the human microbiome. It also lays the foundation for future in vivo studies to determine whether habitat modification by C. accolens could be promoted to control pathogen colonization.
Copyright © 2016 Bomar et al.
Figures


Comment in
-
Unraveling the Molecular Mechanisms Underlying the Nasopharyngeal Bacterial Community Structure.mBio. 2016 Feb 2;7(1):e00009-16. doi: 10.1128/mBio.00009-16. mBio. 2016. PMID: 26838716 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
- R01 AI101018/AI/NIAID NIH HHS/United States
- DE020751/DE/NIDCR NIH HHS/United States
- AI101018/AI/NIAID NIH HHS/United States
- R01 AT007830/AT/NCCIH NIH HHS/United States
- AI066304/AI/NIAID NIH HHS/United States
- R01 AI066304/AI/NIAID NIH HHS/United States
- UL1 RR025758/RR/NCRR NIH HHS/United States
- DE007327/DE/NIDCR NIH HHS/United States
- 1 UL1 RR025758-02/RR/NCRR NIH HHS/United States
- AT007830/AT/NCCIH NIH HHS/United States
- P30 DE020751/DE/NIDCR NIH HHS/United States
- T32 DE007327/DE/NIDCR NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

