Treatment with selectin blocking antibodies after lengthening contractions of mouse muscle blunts neutrophil accumulation but does not reduce damage
- PMID: 26733249
- PMCID: PMC4760404
- DOI: 10.14814/phy2.12667
Treatment with selectin blocking antibodies after lengthening contractions of mouse muscle blunts neutrophil accumulation but does not reduce damage
Abstract
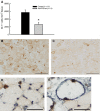
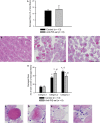
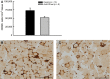
P- and E-selectins are expressed on the surface of endothelial cells and may contribute to neutrophil recruitment following injurious lengthening contractions of skeletal muscle. Blunting neutrophil, but not macrophage, accumulation after lengthening contractions may provide a therapeutic benefit as neutrophils exacerbate damage to muscle fibers, while macrophages promote repair. In this study, we tested the hypothesis that P- and E-selectins contribute to neutrophil, but not macrophage, accumulation in muscles after contraction-induced injury, and that reducing neutrophil accumulation by blocking the selectins would be sufficient to reduce damage to muscle fibers. To test our hypothesis, we treated mice with antibodies to block P- and E-selectin function and assessed leukocyte accumulation and damage in muscles 2 days after lengthening contractions. Treatment with P/E-selectin blocking antibodies reduced neutrophil content by about half in muscles subjected to lengthening contractions. In spite of the reduction in neutrophil accumulation, we did not detect a decrease in damage 2 days after lengthening contractions. We conclude that P- and/or E-selectin contribute to the neutrophil accumulation associated with contraction-induced muscle damage and that only a portion of the neutrophils that typically accumulate following injurious lengthening contractions is sufficient to induce muscle fiber damage and force deficits. Thus, therapeutic interventions based on blocking the selectins or other adhesion proteins will have to reduce neutrophil numbers by more than 50% in order to provide a benefit.
Keywords: Injury; lengthening contraction; muscle; neutrophil; selectin.
© 2015 The Authors. Physiological Reports published by Wiley Periodicals, Inc. on behalf of the American Physiological Society and The Physiological Society.
Figures




References
-
- Baker, W. , Schneider B. A., Kulkarni A., Sloan G., Schaub R., Sypek J., et al. 2004. P‐selectin inhibition suppresses muscle regeneration following injury. J. Leukoc. Biol. 76:352–358. - PubMed
-
- Belcastro, A. N. , Shewchuk L. D., and Raj D. A.. 1998. Exercise‐induced muscle injury: a calpain hypothesis. Mol. Cell. Biochem. 179:135–145. - PubMed
-
- Bosse, R. , and Vestweber D.. 1994. Only simultaneous blocking of the L‐ and P‐selectin completely inhibits neutrophil migration into mouse peritoneum. Eur. J. Immunol. 24:3019–3024. - PubMed
-
- Brickson, S. , Ji L. L., Schell K., Olabisi R., St Pierre Schneider B., and Best T. M.. 2003. M1/70 attenuates blood‐borne neutrophil oxidants, activation, and myofiber damage following stretch injury. J. Appl. Physiol. 95:969–976. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

