The Phytohormone Ethylene Enhances Cellulose Production, Regulates CRP/FNRKx Transcription and Causes Differential Gene Expression within the Bacterial Cellulose Synthesis Operon of Komagataeibacter (Gluconacetobacter) xylinus ATCC 53582
- PMID: 26733991
- PMCID: PMC4686702
- DOI: 10.3389/fmicb.2015.01459
The Phytohormone Ethylene Enhances Cellulose Production, Regulates CRP/FNRKx Transcription and Causes Differential Gene Expression within the Bacterial Cellulose Synthesis Operon of Komagataeibacter (Gluconacetobacter) xylinus ATCC 53582
Abstract
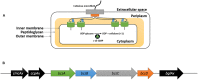
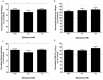
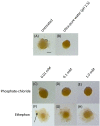
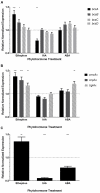
Komagataeibacter (formerly Gluconacetobacter) xylinus ATCC 53582 is a plant-associated model organism for bacterial cellulose (BC) biosynthesis. This bacterium inhabits the carposphere where it interacts with fruit through the bi-directional transfer of phytohormones. The majority of research regarding K. xylinus has been focused on identifying and characterizing structural and regulatory factors that control BC biosynthesis, but its ecophysiology has been generally overlooked. Ethylene is a phytohormone that regulates plant development in a variety of ways, but is most commonly known for its positive role on fruit ripening. In this study, we utilized ethephon (2-chloroethylphosphonic acid) to produce in situ ethylene to investigate the effects of this phytohormone on BC production and the expression of genes known to be involved in K. xylinus BC biosynthesis (bcsA, bcsB, bcsC, bcsD, cmcAx, ccpAx and bglAx). Using pellicle assays and reverse transcription quantitative polymerase chain reaction (RT-qPCR), we demonstrate that ethephon-derived ethylene enhances BC directly in K. xylinus by up-regulating the expression of bcsA and bcsB, and indirectly though the up-regulation of cmcAx, ccpAx, and bglAx. We confirm that IAA directly decreases BC biosynthesis by showing that IAA down-regulates bcsA expression. Similarly, we confirm that ABA indirectly influences BC biosynthesis by showing it does not affect the expression of bcs operon genes. In addition, we are the first to report the ethylene and indole-3-acetic acid (IAA) induced differential expression of genes within the bacterial cellulose synthesis (bcs) operon. Using bioinformatics we have identified a novel phytohormone-regulated CRP/FNRKx transcription factor and provide evidence that it influences BC biosynthesis in K. xylinus. Lastly, utilizing current and previous data, we propose a model for the phytohormone-mediated fruit-bacteria interactions that K. xylinus experiences in nature.
Keywords: CRP/FNR; Komagataeibacter (Gluconacetobacter) xylinus; abscisic acid (ABA); bacterial cellulose; ethylene; fruit-bacteria interaction; indole-3-acetic acid (IAA); plant-microbe interaction.
Figures


Similar articles
-
Complete genome analysis of Gluconacetobacter xylinus CGMCC 2955 for elucidating bacterial cellulose biosynthesis and metabolic regulation.Sci Rep. 2018 Apr 19;8(1):6266. doi: 10.1038/s41598-018-24559-w. Sci Rep. 2018. PMID: 29674724 Free PMC article.
-
Identification and characterization of non-cellulose-producing mutants of Gluconacetobacter hansenii generated by Tn5 transposon mutagenesis.J Bacteriol. 2013 Nov;195(22):5072-83. doi: 10.1128/JB.00767-13. Epub 2013 Sep 6. J Bacteriol. 2013. PMID: 24013627 Free PMC article.
-
Enhancing bacterial cellulose production with hypoxia-inducible factors.Appl Microbiol Biotechnol. 2022 Nov;106(21):7099-7112. doi: 10.1007/s00253-022-12192-7. Epub 2022 Oct 3. Appl Microbiol Biotechnol. 2022. PMID: 36184690
-
Establishing a Role for Bacterial Cellulose in Environmental Interactions: Lessons Learned from Diverse Biofilm-Producing Proteobacteria.Front Microbiol. 2015 Nov 17;6:1282. doi: 10.3389/fmicb.2015.01282. eCollection 2015. Front Microbiol. 2015. PMID: 26635751 Free PMC article. Review.
-
From Nature to Lab: Sustainable Bacterial Cellulose Production and Modification with Synthetic Biology.Polymers (Basel). 2023 Aug 18;15(16):3466. doi: 10.3390/polym15163466. Polymers (Basel). 2023. PMID: 37631523 Free PMC article. Review.
Cited by
-
The power of unbiased phenotypic screens - cellulose as a first receptor for the Schitoviridae phage S6 of Erwinia amylovora.Environ Microbiol. 2022 Aug;24(8):3316-3321. doi: 10.1111/1462-2920.16010. Epub 2022 Apr 19. Environ Microbiol. 2022. PMID: 35415924 Free PMC article.
-
Molecular aspects of bacterial nanocellulose biosynthesis.Microb Biotechnol. 2019 Jul;12(4):633-649. doi: 10.1111/1751-7915.13386. Epub 2019 Mar 18. Microb Biotechnol. 2019. PMID: 30883026 Free PMC article. Review.
-
Plant Growth Promotion Under Water: Decrease of Waterlogging-Induced ACC and Ethylene Levels by ACC Deaminase-Producing Bacteria.Front Microbiol. 2018 May 25;9:1096. doi: 10.3389/fmicb.2018.01096. eCollection 2018. Front Microbiol. 2018. PMID: 29887854 Free PMC article. Review.
-
Ethylene and 1-Aminocyclopropane-1-carboxylate (ACC) in Plant-Bacterial Interactions.Front Plant Sci. 2018 Feb 22;9:114. doi: 10.3389/fpls.2018.00114. eCollection 2018. Front Plant Sci. 2018. PMID: 29520283 Free PMC article. Review.
-
Towards control of cellulose biosynthesis by Komagataeibacter using systems-level and strain engineering strategies: current progress and perspectives.Appl Microbiol Biotechnol. 2020 Aug;104(15):6565-6585. doi: 10.1007/s00253-020-10671-3. Epub 2020 Jun 11. Appl Microbiol Biotechnol. 2020. PMID: 32529377 Free PMC article. Review.
References
-
- Agravante J. U., Matsui T., Kitagawa H. (1990). Starch breakdown and changes in amylase activity during ripening of ethylene and ethanol treated bananas. Acta Hort. 269 133–140. 10.17660/ActaHortic.1990.269.18 - DOI
-
- Arkhipova T. N., Veselov S. U., Melentiev A. I., Martynenko E. V., Kudoyarova G. R. (2005). Ability of bacterium Bacillus subtilis to produce cytokinins and to influence the growth and endogenous hormone content of lettuce plants. Plant Soil 272 201–209. 10.1007/s11104-004-5047-x - DOI
-
- Baca B. E., Elmerich C. (2007). “Microbial production of plant hormones,” in Associative and Endophytic Nitrogen-Fixing Bacteria and Cyanobacterial Associations eds Elmerich C., Newton W. E. (Amsterdam: Springer; ) 113–143. 10.1007/1-4020-3546-2 - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

