Intratracheal Administration of Prostacyclin Analogue-incorporated Nanoparticles Ameliorates the Development of Monocrotaline and Sugen-Hypoxia-induced Pulmonary Arterial Hypertension
- PMID: 26745002
- PMCID: PMC4827325
- DOI: 10.1097/FJC.0000000000000352
Intratracheal Administration of Prostacyclin Analogue-incorporated Nanoparticles Ameliorates the Development of Monocrotaline and Sugen-Hypoxia-induced Pulmonary Arterial Hypertension
Abstract
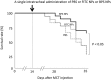
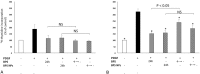
Nanoparticles (NPs) have been used as novel drug delivery systems. Drug-incorporated NPs for local delivery might optimize the efficacy and minimize the side effects of drugs. Intravenous prostacyclin improves long-term survival in patients with pulmonary arterial hypertension (PAH), but it causes serious side effects such as catheter-related infections. We investigated the efficacy and safety of intratracheal administration of a prostacyclin analogue, beraprost (BPS), incorporated NPs in Sugen-hypoxia-normoxia and monocrotaline rat models of PAH and in human PAH pulmonary arterial smooth muscle cells (PASMCs). After a single administration, BPS NPs significantly decreased right ventricular pressure, right ventricular hypertrophy, and pulmonary artery muscularization in the 2 rat models. BPS NPs significantly improved the survival rate in the monocrotaline rat model. No infiltration of inflammatory cells, hemorrhage, or fibrosis was found in the liver, kidney, spleen, and heart after the administration of BPS NPs. No liver or kidney dysfunction was found in the blood examinations. BPS and BPS NPs significantly inhibited the proliferation of human PAH PASMCs after 24 hours of treatment. BPS NPs significantly continued to inhibit the proliferation of human PAH PASMCs at 24 hours after the removal of BPS NPs. BPS NPs significantly induced apoptosis in PAH PASMCs compared to that in non-PAH PASMCs. Intratracheal administration of BPS NPs ameliorates pulmonary hypertension in PAH rat models by a sustained antiproliferative effect and a proapoptotic effect on PAH PASMCs.
Conflict of interest statement
The authors report no conflicts of interest.
Figures
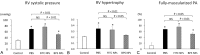
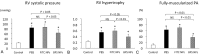




References
-
- Humbert M, Lau EM, Montani D, et al. Advances in therapeutic interventions for patients with pulmonary arterial hypertension. Circulation. 2014;130:2189–2208. - PubMed
-
- Moncada S, Gryglewski R, Bunting S, et al. An enzyme isolated from arteries transforms prostaglandin endoperoxides to an unstable substance that inhibits platelet aggregation. Nature. 1976;263:663–665. - PubMed
-
- Starling MB, Neutze JM, Elliott RL. Control of elevated pulmonary vascular resistance in neonatal swine with prostacyclin (pgi2). Prostaglandins Med. 1979;3:105–117. - PubMed
-
- Rubin LJ, Mendoza J, Hood M, et al. Treatment of primary pulmonary hypertension with continuous intravenous prostacyclin (epoprostenol). Results of a randomized trial. Ann Intern Med. 1990;112:485–491. - PubMed
-
- Barst RJ, Rubin LJ, Long WA, et al. ; Primary Pulmonary Hypertension Study Group. A comparison of continuous intravenous epoprostenol (prostacyclin) with conventional therapy for primary pulmonary hypertension. N Engl J Med. 1996;334:296–301. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

