Diurnal Variations of Circulating Extracellular Vesicles Measured by Nano Flow Cytometry
- PMID: 26745887
- PMCID: PMC4706300
- DOI: 10.1371/journal.pone.0144678
Diurnal Variations of Circulating Extracellular Vesicles Measured by Nano Flow Cytometry
Abstract
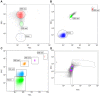
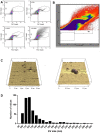
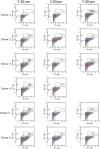
The identification of extracellular vesicles (EVs) as intercellular conveyors of biological information has recently emerged as a novel paradigm in signaling, leading to the exploitation of EVs and their contents as biomarkers of various diseases. However, whether there are diurnal variations in the size, number, and tissue of origin of blood EVs is currently not known, and could have significant implications when using EVs as biomarkers for disease progression. Currently available technologies for the measurement of EV size and number are either time consuming, require specialized equipment, or lack sufficient accuracy across a range of EV sizes. Flow cytometry represents an attractive alternative to these methods; however, traditional flow cytometers are only capable of measuring particles down to 500 nm, which is significantly larger than the average and median sizes of plasma EVs. Utilizing a Beckman Coulter MoFlo XDP flow cytometer with NanoView module, we employed nanoscale flow cytometry (termed nanoFCM) to examine the relative number and scatter distribution of plasma EVs at three different time points during the day in 6 healthy adults. Analysis of liposomes and plasma EVs proved that nanoFCM is capable of detecting biologically-relevant vesicles down to 100 nm in size. With this high resolution configuration, we observed variations in the relative size (FSC/SSC distributions) and concentration (proportions) of EVs in healthy adult plasma across the course of a day, suggesting that there are diurnal variations in the number and size distribution of circulating EV populations. The use of nanoFCM provides a valuable tool for the study of EVs in both health and disease; however, additional refinement of nanoscale flow cytometric methods is needed for use of these instruments for quantitative particle counting and sizing. Furthermore, larger scale studies are necessary to more clearly define the diurnal variations in circulating EVs, and thus further inform their use as biomarkers for disease.
Conflict of interest statement
Figures
Similar articles
-
Assessment of technical and clinical utility of a bead-based flow cytometry platform for multiparametric phenotyping of CNS-derived extracellular vesicles.Cell Commun Signal. 2023 Oct 6;21(1):276. doi: 10.1186/s12964-023-01308-9. Cell Commun Signal. 2023. PMID: 37803478 Free PMC article.
-
Immunoaffinity based methods are superior to kits for purification of prostate derived extracellular vesicles from plasma samples.Prostate. 2017 May;77(13):1335-1343. doi: 10.1002/pros.23393. Epub 2017 Aug 1. Prostate. 2017. PMID: 28762517
-
Prerequisites for the analysis and sorting of extracellular vesicle subpopulations by high-resolution flow cytometry.Cytometry A. 2016 Feb;89(2):135-47. doi: 10.1002/cyto.a.22644. Epub 2015 Feb 16. Cytometry A. 2016. PMID: 25688721
-
Methods to Analyze EVs.Methods Mol Biol. 2017;1545:1-20. doi: 10.1007/978-1-4939-6728-5_1. Methods Mol Biol. 2017. PMID: 27943203 Review.
-
Reliable measurements of extracellular vesicles by clinical flow cytometry.Am J Reprod Immunol. 2021 Feb;85(2):e13350. doi: 10.1111/aji.13350. Epub 2020 Oct 8. Am J Reprod Immunol. 2021. PMID: 32966654 Free PMC article. Review.
Cited by
-
Importance of between and within Subject Variability in Extracellular Vesicle Abundance and Cargo when Performing Biomarker Analyses.Cells. 2021 Feb 24;10(3):485. doi: 10.3390/cells10030485. Cells. 2021. PMID: 33668220 Free PMC article.
-
High-Sensitivity Glycan Profiling of Blood-Derived Immunoglobulin G, Plasma, and Extracellular Vesicle Isolates with Capillary Zone Electrophoresis-Mass Spectrometry.Anal Chem. 2021 Feb 2;93(4):1991-2002. doi: 10.1021/acs.analchem.0c03102. Epub 2021 Jan 12. Anal Chem. 2021. PMID: 33433994 Free PMC article.
-
Flow Cytometric Analysis of Extracellular Vesicles.Methods Mol Biol. 2017;1545:215-225. doi: 10.1007/978-1-4939-6728-5_16. Methods Mol Biol. 2017. PMID: 27943218 Free PMC article.
-
Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines.J Extracell Vesicles. 2018 Nov 23;7(1):1535750. doi: 10.1080/20013078.2018.1535750. eCollection 2018. J Extracell Vesicles. 2018. PMID: 30637094 Free PMC article.
-
Technologies and Standardization in Research on Extracellular Vesicles.Trends Biotechnol. 2020 Oct;38(10):1066-1098. doi: 10.1016/j.tibtech.2020.05.012. Epub 2020 Jun 18. Trends Biotechnol. 2020. PMID: 32564882 Free PMC article. Review.
References
-
- Borges FT, Reis LA, Schor N. Extracellular vesicles: structure, function, and potential clinical uses in renal diseases. Brazilian journal of medical and biological research = Revista brasileira de pesquisas medicas e biologicas / Sociedade Brasileira de Biofisica [et al]. 2013;46(10):824–30. 10.1590/1414-431X20132964 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

