Intravenous Lipid Infusion Induces Endoplasmic Reticulum Stress in Endothelial Cells and Blood Mononuclear Cells of Healthy Adults
- PMID: 26755554
- PMCID: PMC4859375
- DOI: 10.1161/JAHA.115.002574
Intravenous Lipid Infusion Induces Endoplasmic Reticulum Stress in Endothelial Cells and Blood Mononuclear Cells of Healthy Adults
Abstract
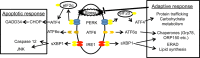
Background: Endoplasmic reticulum (ER) stress and the subsequent unfolded protein response may initially be protective, but when prolonged, have been implicated in atherogenesis in diabetic conditions. Triglycerides and free fatty acids (FFAs) are elevated in patients with diabetes and may contribute to ER stress. We sought to evaluate the effect of acute FFA elevation on ER stress in endothelial and circulating white cells.
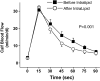
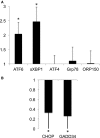
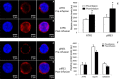
Methods and results: Twenty-one healthy subjects were treated with intralipid (20%; 45 mL/h) plus heparin (12 U/kg/h) infusion for 5 hours. Along with increased triglyceride and FFA levels, intralipid/heparin infusion reduced the calf reactive hyperemic response without a change in conduit artery flow-mediated dilation consistent with microvascular dysfunction. To investigate the short-term effects of elevated triglycerides and FFA, we measured markers of ER stress in peripheral blood mononuclear cells (PBMCs) and vascular endothelial cells (VECs). In VECs, activating transcription factor 6 (ATF6) and phospho-inositol requiring kinase 1 (pIRE1) proteins were elevated after infusion (both P<0.05). In PBMCs, ATF6 and spliced X-box-binding protein 1 (XBP-1) gene expression increased by 2.0- and 2.5-fold, respectively (both P<0.05), whereas CHOP and GADD34 decreased by ≈67% and 74%, respectively (both P<0.01). ATF6 and pIRE1 protein levels also increased (both P<0.05), and confocal microscopy revealed the nuclear localization of ATF6 after infusion, suggesting activation.
Conclusions: Along with microvascular dysfunction, intralipid infusion induced an early protective ER stress response evidenced by activation of ATF6 and IRE1 in both leukocytes and endothelial cells. Our results suggest a potential link between metabolic disturbances and ER stress that may be relevant to vascular disease.
Keywords: endoplasmic reticulum stress; endothelium; free fatty acids; leukocyte.
© 2016 The Authors. Published on behalf of the American Heart Association, Inc., by Wiley Blackwell.
Figures

Similar articles
-
Mechanism of the induction of endoplasmic reticulum stress by the anti-cancer agent, di-2-pyridylketone 4,4-dimethyl-3-thiosemicarbazone (Dp44mT): Activation of PERK/eIF2α, IRE1α, ATF6 and calmodulin kinase.Biochem Pharmacol. 2016 Jun 1;109:27-47. doi: 10.1016/j.bcp.2016.04.001. Epub 2016 Apr 6. Biochem Pharmacol. 2016. PMID: 27059255
-
ZIKV infection activates the IRE1-XBP1 and ATF6 pathways of unfolded protein response in neural cells.J Neuroinflammation. 2018 Sep 21;15(1):275. doi: 10.1186/s12974-018-1311-5. J Neuroinflammation. 2018. PMID: 30241539 Free PMC article.
-
ER stress signaling has an activating transcription factor 6α (ATF6)-dependent "off-switch".J Biol Chem. 2018 Nov 23;293(47):18270-18284. doi: 10.1074/jbc.RA118.002121. Epub 2018 Oct 4. J Biol Chem. 2018. PMID: 30287689 Free PMC article.
-
Endoplasmic Reticulum-Associated Biomarkers for Molecular Phenotyping of Rare Kidney Disease.Int J Mol Sci. 2021 Feb 22;22(4):2161. doi: 10.3390/ijms22042161. Int J Mol Sci. 2021. PMID: 33671535 Free PMC article. Review.
-
Molecular signal networks and regulating mechanisms of the unfolded protein response.J Zhejiang Univ Sci B. 2017 Jan.;18(1):1-14. doi: 10.1631/jzus.B1600043. J Zhejiang Univ Sci B. 2017. PMID: 28070992 Free PMC article. Review.
Cited by
-
Gene Expression of Protein Tyrosine Phosphatase 1B and Endoplasmic Reticulum Stress During Septic Shock.Front Med (Lausanne). 2019 Nov 1;6:240. doi: 10.3389/fmed.2019.00240. eCollection 2019. Front Med (Lausanne). 2019. PMID: 31737637 Free PMC article.
-
Endothelial Cell Metabolism in Atherosclerosis.Front Cell Dev Biol. 2018 Aug 7;6:82. doi: 10.3389/fcell.2018.00082. eCollection 2018. Front Cell Dev Biol. 2018. PMID: 30131957 Free PMC article. Review.
-
Modulation of endothelium function by fatty acids.Mol Cell Biochem. 2022 Jan;477(1):15-38. doi: 10.1007/s11010-021-04260-9. Epub 2021 Sep 16. Mol Cell Biochem. 2022. PMID: 34529222 Free PMC article. Review.
-
The short-chain fatty acid propionate prevents ox-LDL-induced coronary microvascular dysfunction by alleviating endoplasmic reticulum stress in HCMECs.PLoS One. 2024 May 30;19(5):e0304551. doi: 10.1371/journal.pone.0304551. eCollection 2024. PLoS One. 2024. PMID: 38814895 Free PMC article.
-
Effect of endoplasmic reticulum stress on endothelial ischemia-reperfusion injury in humans.Am J Physiol Regul Integr Comp Physiol. 2020 Dec 1;319(6):R666-R672. doi: 10.1152/ajpregu.00257.2020. Epub 2020 Oct 14. Am J Physiol Regul Integr Comp Physiol. 2020. PMID: 33074709 Free PMC article.
References
-
- Steinberg HO, Paradisi G, Hook G, Crowder K, Cronin J, Baron AD. Free fatty acid elevation impairs insulin‐mediated vasodilation and nitric oxide production. Diabetes. 2000;49:1231–1238. - PubMed
-
- Gokce N, Duffy SJ, Hunter LM, Keaney JF Jr, Vita JA. Acute hypertriglyceridemia is associated with peripheral vasodilation and increased basal flow in healthy young adults. Am J Cardiol. 2001;88:153–159. - PubMed
-
- Tripathy D, Mohanty P, Dhindsa S, Syed T, Ghanim H, Aljada A, Dandona P. Elevation of free fatty acids induces inflammation and impairs vascular reactivity in healthy subjects. Diabetes. 2003;52:2882–2887. - PubMed
-
- Boden G, She P, Mozzoli M, Cheung P, Gumireddy K, Reddy P, Xiang X, Luo Z, Ruderman N. Free fatty acids produce insulin resistance and activate the proinflammatory nuclear factor‐kappaB pathway in rat liver. Diabetes. 2005;54:3458–3465. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- R01 HL114675/HL/NHLBI NIH HHS/United States
- HL081587/HL/NHLBI NIH HHS/United States
- T32 HL007224/HL/NHLBI NIH HHS/United States
- R01 HL115391/HL/NHLBI NIH HHS/United States
- HL083801/HL/NHLBI NIH HHS/United States
- K12 HL083781/HL/NHLBI NIH HHS/United States
- R01 HL102299/HL/NHLBI NIH HHS/United States
- HL102299/HL/NHLBI NIH HHS/United States
- HL75795/HL/NHLBI NIH HHS/United States
- HL083269/HL/NHLBI NIH HHS/United States
- HL115775/HL/NHLBI NIH HHS/United States
- P01 HL081587/HL/NHLBI NIH HHS/United States
- HL115391/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

