Chain Walking of Allylrhodium Species Towards Esters During Rhodium-Catalyzed Nucleophilic Allylations of Imines
- PMID: 26756445
- PMCID: PMC4736453
- DOI: 10.1002/anie.201508964
Chain Walking of Allylrhodium Species Towards Esters During Rhodium-Catalyzed Nucleophilic Allylations of Imines
Abstract
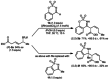
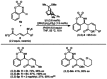
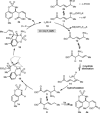
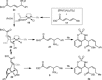
Allylrhodium species derived from δ-trifluoroboryl β,γ-unsaturated esters undergo chain walking towards the ester moiety. The resulting allylrhodium species react with imines to give products containing two new stereocenters and a Z-alkene. By using a chiral diene ligand, products can be obtained with high enantioselectivities, where a pronounced matched/mismatched effect with the chirality of the allyltrifluoroborate is evident.
Keywords: allyltrifluoroborates; asymmetric catalysis; imines; isomerization; rhodium.
© 2015 The Authors. Published by Wiley-VCH Verlag GmbH & Co. KGaA.
Figures



References
-
- For chain walking polymerization, see:
-
- Guan Z., Cotts P. M., McCord E. F., McLain S. J., Science 1999, 283, 2059–2062; - PubMed
-
- Ittel S. D., Johnson L. K., Brookhart M., Chem. Rev. 2000, 100, 1169–1204; - PubMed
-
- Dong Z., Ye Z., Polym. Chem. 2012, 3, 286–301.
-
- For a review of catalytic alkene isomerization-hydroformylation reactions, see: Vilches-Herrera M., Domke L., Börner A., ACS Catal. 2014, 4, 1706–1724.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

