Optimal Synthetic Glycosylation of a Therapeutic Antibody
- PMID: 26756880
- PMCID: PMC4973692
- DOI: 10.1002/anie.201508723
Optimal Synthetic Glycosylation of a Therapeutic Antibody
Abstract
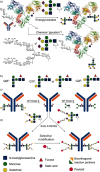
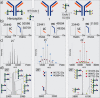
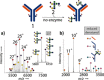
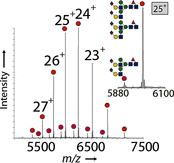
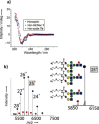
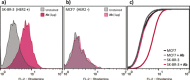
Glycosylation patterns in antibodies critically determine biological and physical properties but their precise control is a significant challenge in biology and biotechnology. We describe herein the optimization of an endoglycosidase-catalyzed glycosylation of the best-selling biotherapeutic Herceptin, an anti-HER2 antibody. Precise MS analysis of the intact four-chain Ab heteromultimer reveals nonspecific, non-enzymatic reactions (glycation), which are not detected under standard denaturing conditions. This competing reaction, which has hitherto been underestimated as a source of side products, can now be minimized. Optimization allowed access to the purest natural form of Herceptin to date (≥90 %). Moreover, through the use of a small library of sugars containing non-natural functional groups, Ab variants containing defined numbers of selectively addressable chemical tags (reaction handles at Sia C1) in specific positions (for attachment of cargo molecules or "glycorandomization") were readily generated.
Keywords: antibodies; endoglycosidases; glycoengineering; glycosylation; native mass spectrometry.
© 2016 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

