α-Synuclein and huntingtin exon 1 amyloid fibrils bind laterally to the cellular membrane
- PMID: 26757959
- PMCID: PMC4725933
- DOI: 10.1038/srep19180
α-Synuclein and huntingtin exon 1 amyloid fibrils bind laterally to the cellular membrane
Abstract
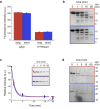
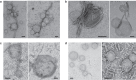
Fibrillar aggregates involved in neurodegenerative diseases have the ability to spread from one cell to another in a prion-like manner. The underlying molecular mechanisms, in particular the binding mode of the fibrils to cell membranes, are poorly understood. In this work we decipher the modality by which aggregates bind to the cellular membrane, one of the obligatory steps of the propagation cycle. By characterizing the binding properties of aggregates made of α-synuclein or huntingtin exon 1 protein displaying similar composition and structure but different lengths to mammalian cells we demonstrate that in both cases aggregates bind laterally to the cellular membrane, with aggregates extremities displaying little or no role in membrane binding. Lateral binding to artificial liposomes was also observed by transmission electron microscopy. In addition we show that although α-synuclein and huntingtin exon 1 fibrils bind both laterally to the cellular membrane, their mechanisms of interaction differ. Our findings have important implications for the development of future therapeutic tools that aim to block protein aggregates propagation in the brain.
Figures




References
-
- Knowles T. P. J., Vendruscolo M. & Dobson C. M. The amyloid state and its association with protein misfolding diseases. Nat. Rev. Mol. Cell Biol. 15, 384–396 (2014). - PubMed
-
- Peelaerts W. et al. α-Synuclein strains cause distinct synucleinopathies after local and systemic administration. Nature 522, 340–344 (2015). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

