Exemestane Use in Postmenopausal Women at High Risk for Invasive Breast Cancer: Evaluating Biomarkers of Efficacy and Safety
- PMID: 26758879
- PMCID: PMC4777638
- DOI: 10.1158/1940-6207.CAPR-15-0269
Exemestane Use in Postmenopausal Women at High Risk for Invasive Breast Cancer: Evaluating Biomarkers of Efficacy and Safety
Abstract
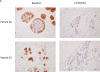
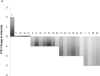
This phase II trial evaluated clinical markers of efficacy and safety of exemestane in postmenopausal women at increased risk for breast cancer. Postmenopausal women (n = 42) at risk for invasive breast cancer received 25 mg exemestane daily for 2 years along with calcium and vitamin D. The primary outcome was change in mammographic density (MD) after one year. Secondary outcomes included change in serum steroid hormones as well as change in trefoil protein 1 (TFF1) and proliferating cell nuclear antigen (PCNA) in breast tissue. Safety and tolerability were also assessed. MD decreased at 1 year and was significant at 2 years [mean change = -4.1%; 95% confidence intervals (CI), -7.2 to -1.1; P = 0.009]. Serum estradiol and testosterone levels significantly decreased at 3 months and remained suppressed at 12 months. After 1 year of treatment, TFF1 intensity decreased (mean change -1.32; 95% CI, -1.87 to -0.76; P < 0.001). Exemestane was safe and well tolerated. Exemestane decreased MD and expression of breast tissue TFF1. It was well tolerated with few clinically relevant side effects. MD and breast tissue TFF1 are potential biomarkers of breast cancer-preventive effects of exemestane in high-risk postmenopausal women.
Trial registration: ClinicalTrials.gov NCT00073073.
©2016 American Association for Cancer Research.
Figures
Similar articles
-
Effects of adjuvant exemestane versus anastrozole on bone mineral density for women with early breast cancer (MA.27B): a companion analysis of a randomised controlled trial.Lancet Oncol. 2014 Apr;15(4):474-82. doi: 10.1016/S1470-2045(14)70035-X. Epub 2014 Mar 11. Lancet Oncol. 2014. PMID: 24636210 Free PMC article. Clinical Trial.
-
The effect of exemestane on serum lipid profile in postmenopausal women with metastatic breast cancer: a companion study to EORTC Trial 10951, 'Randomized phase II study in first line hormonal treatment for metastatic breast cancer with exemestane or tamoxifen in postmenopausal patients'.Ann Oncol. 2004 Feb;15(2):211-7. doi: 10.1093/annonc/mdh064. Ann Oncol. 2004. PMID: 14760111 Clinical Trial.
-
Exemestane for breast-cancer prevention in postmenopausal women.N Engl J Med. 2011 Jun 23;364(25):2381-91. doi: 10.1056/NEJMoa1103507. Epub 2011 Jun 4. N Engl J Med. 2011. PMID: 21639806 Clinical Trial.
-
Exemestane: a review of its use in postmenopausal women with breast cancer.Drugs. 2009;69(7):889-918. doi: 10.2165/00003495-200969070-00007. Drugs. 2009. PMID: 19441873 Review.
-
Reducing the risk for breast cancer recurrence after completion of tamoxifen treatment in postmenopausal women.Clin Ther. 2007 Aug;29(8):1535-47. doi: 10.1016/j.clinthera.2007.08.013. Clin Ther. 2007. PMID: 17919537 Review.
Cited by
-
Drug repurposing for cancer therapy.Signal Transduct Target Ther. 2024 Apr 19;9(1):92. doi: 10.1038/s41392-024-01808-1. Signal Transduct Target Ther. 2024. PMID: 38637540 Free PMC article. Review.
-
Variation in the UGT2B17 genotype, exemestane metabolism and menopause-related toxicities in the CCTG MAP.3 trial.Breast Cancer Res Treat. 2020 Oct;183(3):705-716. doi: 10.1007/s10549-020-05812-1. Epub 2020 Aug 1. Breast Cancer Res Treat. 2020. PMID: 32715442 Free PMC article.
-
Aromatase Inhibitors and Plasma Lipid Changes in Postmenopausal Women with Breast Cancer: A Systematic Review and Meta-Analysis.J Clin Med. 2024 Mar 21;13(6):1818. doi: 10.3390/jcm13061818. J Clin Med. 2024. PMID: 38542042 Free PMC article. Review.
-
Transcriptome analysis of endometrial tissues following GnRH agonist treatment in a mouse adenomyosis model.Drug Des Devel Ther. 2017 Mar 9;11:695-704. doi: 10.2147/DDDT.S127889. eCollection 2017. Drug Des Devel Ther. 2017. PMID: 28331289 Free PMC article.
-
Mammographic density: a potential monitoring biomarker for adjuvant and preventative breast cancer endocrine therapies.Oncotarget. 2017 Jan 17;8(3):5578-5591. doi: 10.18632/oncotarget.13484. Oncotarget. 2017. PMID: 27894075 Free PMC article. Review.
References
-
- Goss PE, Ingle JN, Ales-Martinez JE, Cheung AM, Chlebowski RT, Wactawski-Wende J, et al. Exemestane for breast-cancer prevention in postmenopausal women. N Engl J Med. 2011;364:2381–91. - PubMed
-
- Cuzick J, Powles T, Veronesi U, Forbes J, Edwards R, Ashley S, et al. Overview of the main outcomes in breast-cancer prevention trials. Lancet. 2003;361:296–300. - PubMed
-
- Visvanathan K, Hurley P, Bantug E, Brown P, Col NF, Cuzick J, et al. Use of pharmacologic interventions for breast cancer risk reduction: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2013;31:2942–62. - PubMed
-
- Cuzick J, Sestak I, Forbes JF, Dowsett M, Knox J, Cawthorn S, et al. Anastrozole for prevention of breast cancer in high-risk postmenopausal women (IBIS-II): an international, double-blind, randomised placebo-controlled trial. Lancet. 2014;383:1041–8. - PubMed
-
- Boyd NF, Byng JW, Jong RA, Fishell EK, Little LE, Miller AB, et al. Quantitative classification of mammographic densities and breast cancer risk: results from the Canadian National Breast Screening Study. J Natl Cancer Inst. 1995;87:670–5. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

