Thermal Destabilization of Collagen Matrix Hierarchical Structure by Freeze/Thaw
- PMID: 26765741
- PMCID: PMC4713088
- DOI: 10.1371/journal.pone.0146660
Thermal Destabilization of Collagen Matrix Hierarchical Structure by Freeze/Thaw
Abstract
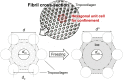
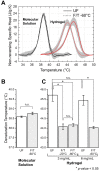
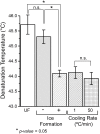
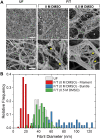
This study aims to characterize and understand the effects of freezing on collagen structures and functionality. Specifically, thermodynamic destabilization of collagen at molecular- and fibril-levels by combination of low temperatures and freezing were experimentally characterized using modulated differential scanning calorimetry. In order to delineate the effects of sub-zero temperature and water-ice phase change, we hypothesized that the extent of destabilization can be determined based on post-thaw heat induced thermal denaturation of collagen. It is found that thermal denaturation temperature of collagen in hydrogel decreases by 1.4-1.6°C after freeze/thaw while no such decrease is observed in the case of molecular solution. The destabilization is predominantly due to ice formation. Exposure to low temperatures in the absence of ice has only minimal effect. Calorimetry measurements combined with morphological examination of collagen matrices by scanning electron microscopy suggest that freezing results in destabilization of collagen fibrils due to expansion of intrafibrillar space by ice formation. This fibril-level damage can be alleviated by use of cryoprotectant DMSO at concentrations as low as 0.5 M. A theoretical model explaining the change in collagen post-thaw thermal stability by freezing-induced fibril expansion is also proposed.
Conflict of interest statement
Figures


References
-
- Fratzl P. Collagen: Structure and Mechanics, an Introduction. In: Fratzl P, editor.: Springer; US; 2008. p. 1–13.
-
- Wess TJ. Collagen Fibrillar Structure and Hierarchies. In: Fratzl P, editor.: Springer; US; 2008. p. 49–80.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

