Current Progress in Therapeutic Gene Editing for Monogenic Diseases
- PMID: 26765770
- PMCID: PMC4786935
- DOI: 10.1038/mt.2016.5
Current Progress in Therapeutic Gene Editing for Monogenic Diseases
Abstract
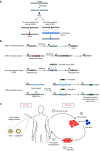
Programmable nucleases allow defined alterations in the genome with ease-of-use, efficiency, and specificity. Their availability has led to accurate and widespread genome engineering, with multiple applications in basic research, biotechnology, and therapy. With regard to human gene therapy, nuclease-based gene editing has facilitated development of a broad range of therapeutic strategies based on both nonhomologous end joining and homology-dependent repair. This review discusses current progress in nuclease-based therapeutic applications for a subset of inherited monogenic diseases including cystic fibrosis, Duchenne muscular dystrophy, diseases of the bone marrow, and hemophilia and highlights associated challenges and future prospects.
Figures
References
-
- In: Rodwell, C., Aymé, S. (eds.) (2014). 2014 Report on the State of the Art of Rare Disease Activities in Europe Part Ii: Key Developments in the Field of Rare Diseases in Europe in 2013. 1–90. http://www.eucerd.eu/upload/file/Reports/2014ReportStateofArtRDActivitie....
-
- Alton, EW, Armstrong, DK, Ashby, D, Bayfield, KJ, Bilton, D, Bloomfield, EV et al.; UK Cystic Fibrosis Gene Therapy Consortium. (2015). Repeated nebulisation of non-viral CFTR gene therapy in patients with cystic fibrosis: a randomised, double-blind, placebo-controlled, phase 2b trial. Lancet Respir Med 3: 684–691. - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

