Antioxidant and Metal Chelation-Based Therapies in the Treatment of Prion Disease
- PMID: 26784872
- PMCID: PMC4665489
- DOI: 10.3390/antiox3020288
Antioxidant and Metal Chelation-Based Therapies in the Treatment of Prion Disease
Abstract
Many neurodegenerative disorders involve the accumulation of multimeric assemblies and amyloid derived from misfolded conformers of constitutively expressed proteins. In addition, the brains of patients and experimental animals afflicted with prion disease display evidence of heightened oxidative stress and damage, as well as disturbances to transition metal homeostasis. Utilising a variety of disease model paradigms, many laboratories have demonstrated that copper can act as a cofactor in the antioxidant activity displayed by the prion protein while manganese has been implicated in the generation and stabilisation of disease-associated conformers. This and other evidence has led several groups to test dietary and chelation therapy-based regimens to manipulate brain metal concentrations in attempts to influence the progression of prion disease in experimental mice. Results have been inconsistent. This review examines published data on transition metal dyshomeostasis, free radical generation and subsequent oxidative damage in the pathogenesis of prion disease. It also comments on the efficacy of trialed therapeutics chosen to combat such deleterious changes.
Keywords: CJD; Cu; Mn; SOD2; amyloid; antioxidant; chelation; hydroxyl radical; oxidative stress; superoxide dismutase; therapy; transmissible spongiform encephalopathy.
Figures
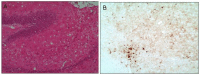
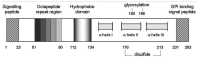

Similar articles
-
Metals in obex and retropharyngeal lymph nodes of Illinois white-tailed deer and their variations associated with CWD status.Prion. 2015;9(1):48-58. doi: 10.1080/19336896.2015.1019194. Prion. 2015. PMID: 25695915 Free PMC article.
-
Does an infrasonic acoustic shock wave resonance of the manganese 3+ loaded/copper depleted prion protein initiate the pathogenesis of TSE?Med Hypotheses. 2003 Jun;60(6):797-820. doi: 10.1016/s0306-9877(03)00007-0. Med Hypotheses. 2003. PMID: 12699706
-
The effects of prion protein expression on metal metabolism.Mol Cell Neurosci. 2009 Jun;41(2):135-47. doi: 10.1016/j.mcn.2009.02.002. Epub 2009 Feb 21. Mol Cell Neurosci. 2009. PMID: 19233277
-
Copper-dependent functions for the prion protein.Mol Biotechnol. 2002 Oct;22(2):165-78. doi: 10.1385/MB:22:2:165. Mol Biotechnol. 2002. PMID: 12405264 Review.
-
Metal Dyshomeostasis and Their Pathological Role in Prion and Prion-Like Diseases: The Basis for a Nutritional Approach.Front Neurosci. 2017 Jan 19;11:3. doi: 10.3389/fnins.2017.00003. eCollection 2017. Front Neurosci. 2017. PMID: 28154522 Free PMC article. Review.
Cited by
-
Carnosic Acid and Carnosol Display Antioxidant and Anti-Prion Properties in In Vitro and Cell-Free Models of Prion Diseases.Antioxidants (Basel). 2022 Apr 6;11(4):726. doi: 10.3390/antiox11040726. Antioxidants (Basel). 2022. PMID: 35453411 Free PMC article.
-
Oxidative Stress and Huntington's Disease: The Good, The Bad, and The Ugly.J Huntingtons Dis. 2016 Oct 1;5(3):217-237. doi: 10.3233/JHD-160205. J Huntingtons Dis. 2016. PMID: 27662334 Free PMC article. Review.
-
Silicon Oxynitrophosphide Nanoscale Coating Enhances Antioxidant Marker-Induced Angiogenesis During in vivo Cranial Bone-Defect Healing.JBMR Plus. 2021 Mar 18;5(4):e10425. doi: 10.1002/jbm4.10425. eCollection 2021 Apr. JBMR Plus. 2021. PMID: 33869985 Free PMC article.
References
-
- Melov S., Doctrow S.R., Schneider J.A., Haberson J., Patel M., Coskun P.E., Huffman K., Wallace D.C., Malfroy B. Lifespan extension and rescue of spongiform encephalopathy in superoxide dismutase 2 nullizygous mice treated with superoxide dismutase-catalase mimetics. J. Neurosci. 2001;21:8348–8353. - PMC - PubMed
-
- Knight R., Brazier M., Collins S. Prions: A challenge for Science, Medicine and Public Health Systems. In: Rabenau H.F., Cinatl J., Doerr H.W., editors. Contributions to Microbiology. Volume 11 Karger; Basel, Switzerland: 2004.
-
- Goldfarb L.G., Petersen R.B., Tabaton M., Brown P., LeBlanc A.C., Montagna P., Cortelli P., Julien J., Vital C., Pendelbury W.W., et al. Fatal familial insomnia and familial Creutzfeldt-Jakob disease: Disease phenotype determined by a DNA polymorphism. Science. 1992;258:806–808. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

