Evidence for a biphasic mode of respiratory syncytial virus transmission in permissive HEp2 cell monolayers
- PMID: 26790623
- PMCID: PMC4719537
- DOI: 10.1186/s12985-016-0467-9
Evidence for a biphasic mode of respiratory syncytial virus transmission in permissive HEp2 cell monolayers
Abstract
Background: During respiratory syncytial virus (RSV) infection filamentous virus particles are formed on the cell surface. Although the virus infectivity remains cell-associated, low levels of cell-free virus is detected during advanced infection. It is currently unclear if this cell-free virus infectivity is due to a low-efficiency specific cell-release mechanism, or if it arises due to mechanical breakage following virus-induced cell damage at the advanced stage of infection. Understanding the origin of this cell-free virus is a prerequisite for understanding the mechanism of RSV transmission in permissive cells. In this study we describe a detailed examination of RSV transmission in permissive HEp2 cell monolayers.
Methods: HEp2 cell monolayers were infected with RSV using a multiplicity of infection of 0.0002, and the course of infection monitored over 5 days. The progression of the virus infection within the cell monolayers was performed using bright-field microscopy to visualise the cell monolayer and immunofluorescence microscopy to detect virus-infected cells. The cell-associated and cell-free virus infectivity were determined by virus plaque assay, and the virus-induced cell cytotoxicity determined by measuring cell membrane permeability and cellular DNA fragmentation.
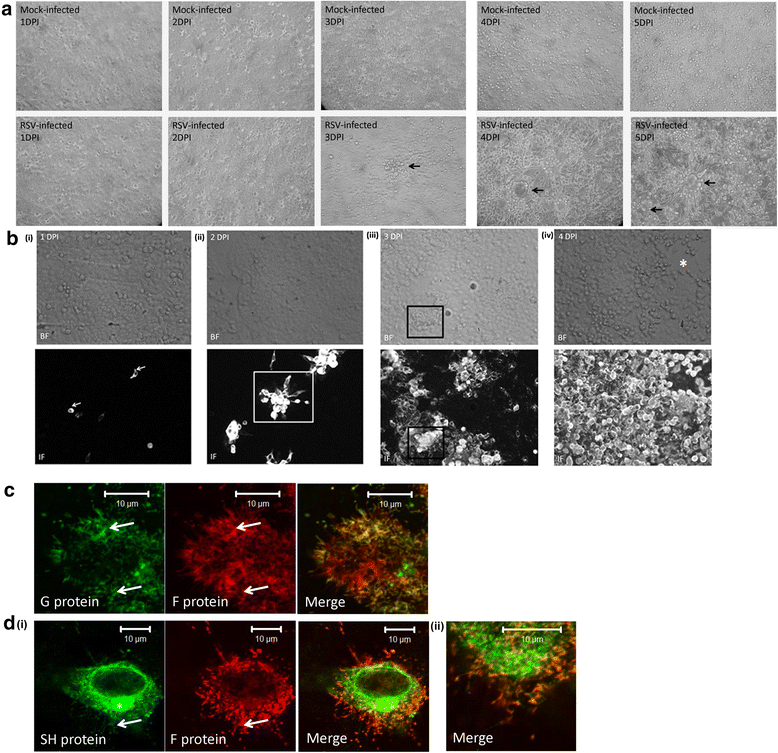
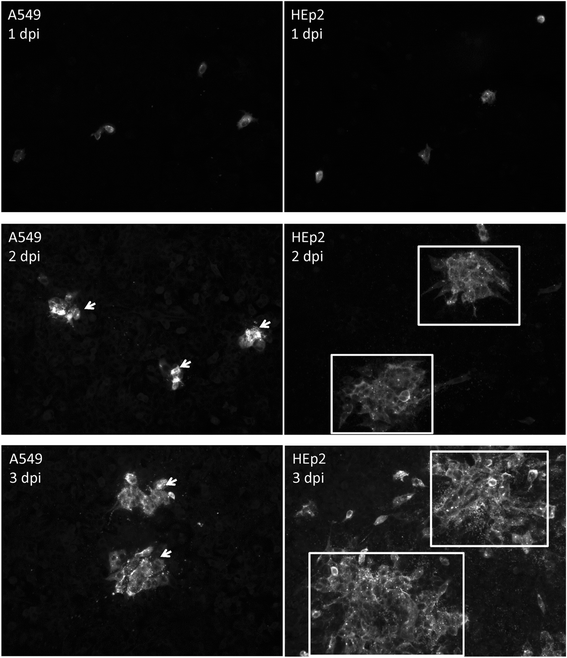
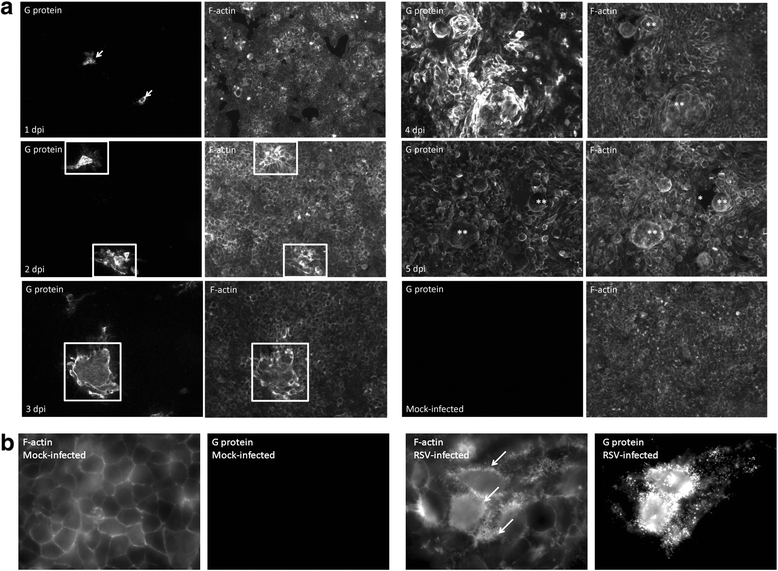
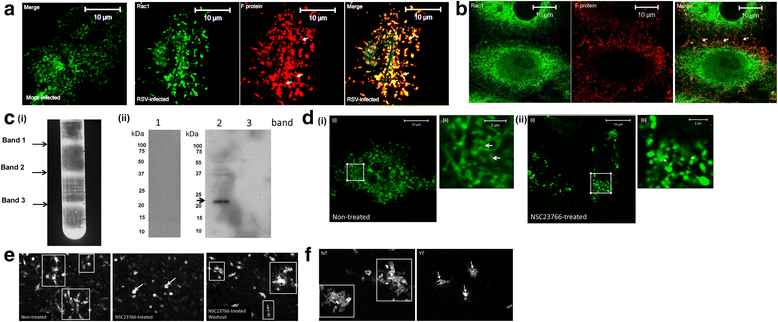
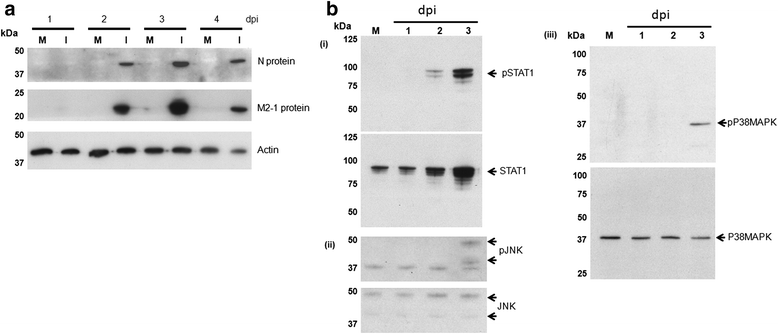
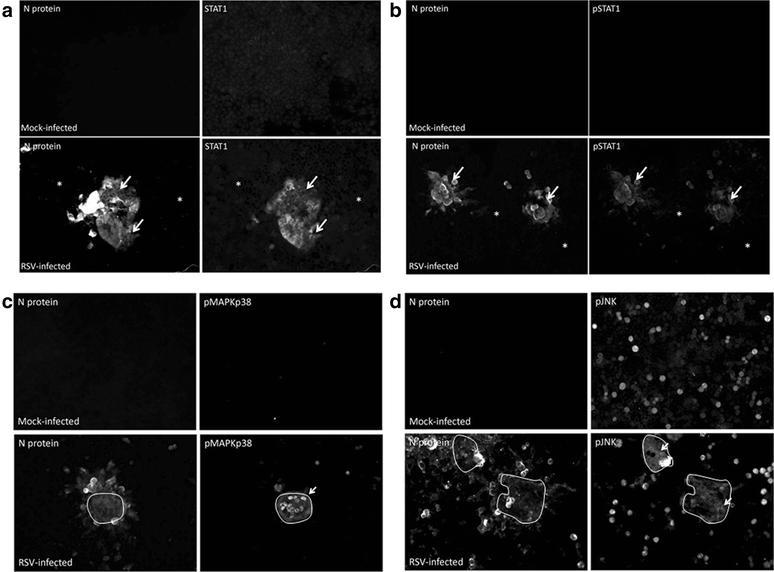
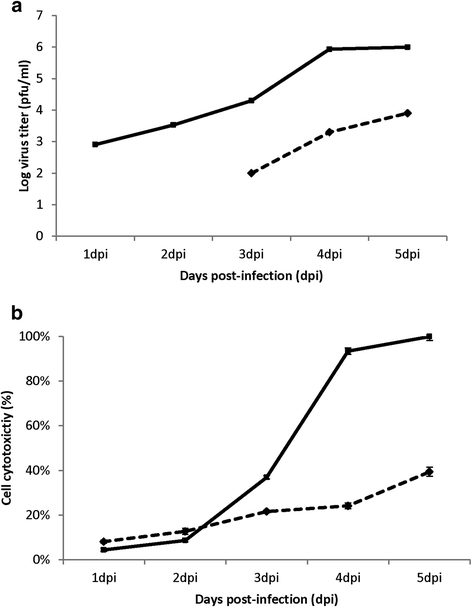
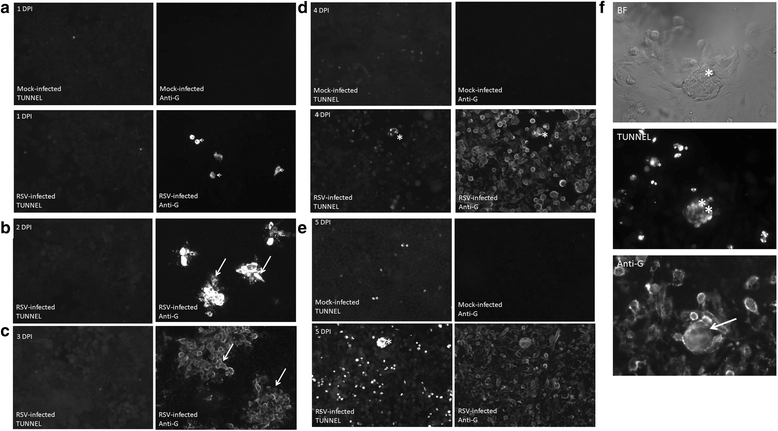
Results: At 2 days-post infection (dpi), large clusters of virus-infected cells could be detected indicating localised transmission in the cell monolayer, and during this stage we failed to detect either cell-free virus or cell cytotoxicity. At 3 dpi the presence of much larger infected cell clusters correlated with the begining of virus-induced changes in cell permeability. The presence of cell-free virus correlated with continued increase in cell permeability and cytotoxicity at 4 and 5 dpi. At 5 dpi extensive cell damage, syncytial formation, and increased cellular DNA fragmentation was noted. However, even at 5 dpi the cell-free virus constituted less than 1 % of the total virus infectivity.
Conclusions: Our data supports a model of RSV transmission that initially involves the localised cell-to-cell spread of virus particles within the HEp2 cell monolayer. However, low levels of cell free-virus infectivity was observed at the advanced stages of infection, which correlated with a general loss in cell monolayer integrity due to virus-induced cytotoxicity.
Figures

Similar articles
-
Viperin protein expression inhibits the late stage of respiratory syncytial virus morphogenesis.Antiviral Res. 2015 Feb;114:11-20. doi: 10.1016/j.antiviral.2014.11.007. Epub 2014 Nov 26. Antiviral Res. 2015. PMID: 25433308
-
The link between respiratory syncytial virus (RSV) morphogenesis and virus transmission: Towards a paradigm for understanding RSV transmission in the upper airway.Virology. 2025 Mar;604:110413. doi: 10.1016/j.virol.2025.110413. Epub 2025 Jan 22. Virology. 2025. PMID: 39869971 Review.
-
Respiratory syncytial virus causes increased bronchial epithelial permeability.Chest. 2004 Jul;126(1):186-91. doi: 10.1378/chest.126.1.186. Chest. 2004. PMID: 15249461
-
Direct Inhibition of Cellular Fatty Acid Synthase Impairs Replication of Respiratory Syncytial Virus and Other Respiratory Viruses.PLoS One. 2015 Dec 11;10(12):e0144648. doi: 10.1371/journal.pone.0144648. eCollection 2015. PLoS One. 2015. PMID: 26659560 Free PMC article.
-
Contribution of neuroimmune mechanisms to airway inflammation and remodeling during and after respiratory syncytial virus infection.Pediatr Infect Dis J. 2003 Feb;22(2 Suppl):S66-74; discussion S74-5. doi: 10.1097/01.inf.0000053888.67311.1d. Pediatr Infect Dis J. 2003. PMID: 12671455 Review.
Cited by
-
The reproduction number and its probability distribution for stochastic viral dynamics.J R Soc Interface. 2024 Jan;21(210):20230400. doi: 10.1098/rsif.2023.0400. Epub 2024 Jan 24. J R Soc Interface. 2024. PMID: 38264928 Free PMC article.
-
A comparison of RSV and influenza in vitro kinetic parameters reveals differences in infecting time.PLoS One. 2018 Feb 8;13(2):e0192645. doi: 10.1371/journal.pone.0192645. eCollection 2018. PLoS One. 2018. PMID: 29420667 Free PMC article.
-
Caveolae provide a specialized membrane environment for respiratory syncytial virus assembly.J Cell Sci. 2017 Mar 15;130(6):1037-1050. doi: 10.1242/jcs.198853. Epub 2017 Feb 2. J Cell Sci. 2017. PMID: 28154158 Free PMC article.
-
Defining the Assembleome of the Respiratory Syncytial Virus.Subcell Biochem. 2023;106:227-249. doi: 10.1007/978-3-031-40086-5_9. Subcell Biochem. 2023. PMID: 38159230 Review.
-
Impaired Nuclear Export of the Ribonucleoprotein Complex and Virus-Induced Cytotoxicity Combine to Restrict Propagation of the A/Duck/Malaysia/02/2001 (H9N2) Virus in Human Airway Cells.Cells. 2020 Feb 3;9(2):355. doi: 10.3390/cells9020355. Cells. 2020. PMID: 32028682 Free PMC article.
References
-
- Nair H, Nokes DJ, Gessner BD, Dherani M, Madhi SA, Singleton RJ, et al. Global burden of acute lower respiratory infections due to respiratory syncytial virus in young children: a systematic review and meta-analysis. The Lancet. 2010;375:1545–55. doi: 10.1016/S0140-6736(10)60206-1. - DOI - PMC - PubMed
-
- Garcia J, Garcia-Barreno B, Vivo A, Melero JA. Cytoplasmic inclusions of respiratory syncytial virus-infected cells: formation of inclusion bodies in transfected cells that coexpress the nucleoprotein, the phosphoprotein, and the 22 K protein. Virology. 1993;195:243–7. doi: 10.1006/viro.1993.1366. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

