Evidence for enhanced collagen type III deposition focally in the territorial matrix of osteoarthritic hip articular cartilage
- PMID: 26790721
- PMCID: PMC4896400
- DOI: 10.1016/j.joca.2016.01.001
Evidence for enhanced collagen type III deposition focally in the territorial matrix of osteoarthritic hip articular cartilage
Abstract
Objective: To determine if type III collagen is concentrated in the chymotrypsin-extractable collagen pool from osteoarthritic articular cartilage to assess its potential as a biomarker of Osteoarthritis (OA) pathogenic mechanisms.
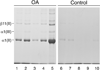
Methods: Full thickness articular cartilage from grossly normal surfaces was analyzed from femoral heads, obtained at hip replacement surgery, from OA (n = 10) and fracture (n = 10) patients. Collagen, extracted by α-chymotrypsin, was characterized by SDS-PAGE/Western blot analysis, ELISA and immunohistochemistry using monoclonal antibodies specific to collagens types II and III.
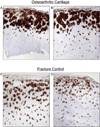
Results: α-Chymotrypsin extracted more collagen from OA than control cartilage. The extractable pool included collagen types II and III from both OA and control hips. Importantly, OA cartilage contained 6-fold more collagen type III than control cartilage, based on ELISA. The estimated total tissue ratio of collagen III/II was in the 1-10% range for individual OA cartilage samples, based on pepsin-solubilized collagen using SDS-PAGE densitometry. Collagen type III N-propeptide trimers were the main molecular fragments seen on Western blot analysis of OA and control extracts. The chymotrypsin-extracted type II collagen gave primarily full-length α1(II) chains and chain fragments of α1(II) on Western blot analysis from both OA and control tissues. Immunohistochemistry showed that type III collagen was more concentrated in the upper half of OA cartilage and in the territorial matrix around individual chondrocytes and chondrocyte clusters.
Conclusions: The findings confirm that collagen type III deposition occurs in adult articular cartilage but significantly more pronounced in osteoarthritic joints, presenting a potential marker of matrix repair or pathobiology.
Keywords: Articular cartilage; Collagen type II and type III; Hip osteoarthritis.
Copyright © 2016 Osteoarthritis Research Society International. Published by Elsevier Ltd. All rights reserved.
Conflict of interest statement
There is no conflict of interest for any of the authors.
Figures


Similar articles
-
Cartilage collagen damage in hip osteoarthritis similar to that seen in knee osteoarthritis; a case-control study of relationship between collagen, glycosaminoglycan and cartilage swelling.BMC Musculoskelet Disord. 2013 Jan 9;14:18. doi: 10.1186/1471-2474-14-18. BMC Musculoskelet Disord. 2013. PMID: 23302451 Free PMC article.
-
Independent expression of fibril-forming collagens I, II, and III in chondrocytes of human osteoarthritic cartilage.J Clin Invest. 1993 Mar;91(3):829-37. doi: 10.1172/JCI116303. J Clin Invest. 1993. PMID: 7680669 Free PMC article.
-
Presence and distribution of collagen II, collagen I, fibronectin, and tenascin in rabbit normal and osteoarthritic cartilage.J Rheumatol. 1999 Feb;26(2):386-94. J Rheumatol. 1999. PMID: 9972974
-
Molecular regulation of articular chondrocyte function and its significance in osteoarthritis.Histol Histopathol. 2011 Mar;26(3):377-94. doi: 10.14670/HH-26.377. Histol Histopathol. 2011. PMID: 21210351 Review.
-
Preserving the longevity of long-lived type II collagen and its implication for cartilage therapeutics.Ageing Res Rev. 2016 Jul;28:62-71. doi: 10.1016/j.arr.2016.04.011. Epub 2016 Apr 28. Ageing Res Rev. 2016. PMID: 27133944 Review.
Cited by
-
Impact of type III collagen on monosodium iodoacetate-induced osteoarthritis in rats.Heliyon. 2020 Jun 7;6(6):e04083. doi: 10.1016/j.heliyon.2020.e04083. eCollection 2020 Jun. Heliyon. 2020. PMID: 32548322 Free PMC article.
-
Effect of Moderate Exercise on the Superficial Zone of Articular Cartilage in Age-Related Osteoarthritis.Diagnostics (Basel). 2023 Oct 12;13(20):3193. doi: 10.3390/diagnostics13203193. Diagnostics (Basel). 2023. PMID: 37892013 Free PMC article.
-
Human Chondrocyte Activation by Toxins From Premolis semirufa, an Amazon Rainforest Moth Caterpillar: Identifying an Osteoarthritis Signature.Front Immunol. 2020 Sep 18;11:2191. doi: 10.3389/fimmu.2020.02191. eCollection 2020. Front Immunol. 2020. PMID: 33072083 Free PMC article.
-
Selective Enzymatic Digestion of Proteoglycans and Collagens Alters Cartilage T1rho and T2 Relaxation Times.Ann Biomed Eng. 2019 Jan;47(1):190-201. doi: 10.1007/s10439-018-02143-7. Epub 2018 Oct 4. Ann Biomed Eng. 2019. PMID: 30288634 Free PMC article.
-
Collagen: quantification, biomechanics, and role of minor subtypes in cartilage.Nat Rev Mater. 2020 Oct;5(10):730-747. doi: 10.1038/s41578-020-0213-1. Epub 2020 Jul 20. Nat Rev Mater. 2020. PMID: 33996147 Free PMC article.
References
-
- Eyre DR, Weis MA, Wu JJ. Articular cartilage collagen: an irreplaceable framework? Eur Cell Mater. 2006;12:57–63. - PubMed
-
- Maroudas A. Adult Articular Cartilage. 2nd. London: Pitman Medical; 1979. Physicochemical properties of articular cartilage.
-
- Dahlberg L, Billinghurst RC, Manner P, Nelson F, Webb G, Ionescu M, et al. Selective enhancement of collagenase-mediated cleavage of resident type II collagen in cultured osteoarthritic cartilage and arrest with a synthetic inhibitor that spares collagenase 1 (matrix metalloproteinase 1) Arthritis Rheum. 2000;43:673–682. - PubMed
-
- Floman Y, Eyre DR, Glimcher MJ. Induction of osteoarthrosis in the rabbit knee joint: biochemical studies on the articular cartilage. Clin Orthop Relat Res. 1980:278–286. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

