Apremilast, an oral phosphodiesterase 4 inhibitor, in patients with psoriatic arthritis and current skin involvement: a phase III, randomised, controlled trial (PALACE 3)
- PMID: 26792812
- PMCID: PMC4893110
- DOI: 10.1136/annrheumdis-2015-207963
Apremilast, an oral phosphodiesterase 4 inhibitor, in patients with psoriatic arthritis and current skin involvement: a phase III, randomised, controlled trial (PALACE 3)
Abstract
Objective: To evaluate apremilast treatment in patients with active psoriatic arthritis, including current skin involvement, despite prior therapy with conventional disease-modifying antirheumatic drugs and/or biologic agents.
Methods: Patients (N=505) were randomised (1:1:1) to placebo, apremilast 20 mg twice daily, or apremilast 30 mg twice daily. Rescue therapy with apremilast was designated at week 16 for placebo patients not achieving 20% improvement in swollen and tender joint counts. At week 24, the remaining placebo patients were then randomised to apremilast 20 mg twice daily or 30 mg twice daily. The efficacy and safety of apremilast were assessed over 52 weeks.
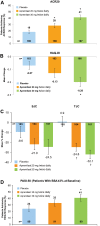
Results: At week 16, significantly more patients receiving apremilast 20 mg twice daily (28%) and 30 mg twice daily (41%) achieved 20% improvement in American College of Rheumatology response criteria versus placebo (18%; p=0.0295 and p<0.0001, respectively), and mean decrease in the Health Assessment Questionnaire-Disability Index score was significantly greater with apremilast 30 mg twice daily (-0.20) versus placebo (-0.07; p=0.0073). In patients with baseline psoriasis body surface area involvement ≥3%, significantly more apremilast 30 mg twice daily patients achieved 50% reduction from baseline Psoriasis Area and Severity Index score (41%) versus placebo (24%; p=0.0098) at week 16. At week 52, observed improvements in these measures demonstrated sustained response with continued apremilast treatment. Most adverse events were mild to moderate in severity; the most common were diarrhoea, nausea, headache and upper respiratory tract infection.
Conclusions: Apremilast demonstrated clinically meaningful improvements in psoriatic arthritis and psoriasis at week 16; sustained improvements were seen with continued treatment through 52 weeks. Apremilast was generally well tolerated and demonstrated an acceptable safety profile.
Trial registration number: NCT01212770.
Keywords: Ankylosing Spondylitis; Psoriatic Arthritis; Treatment.
Published by the BMJ Publishing Group Limited. For permission to use (where not already granted under a licence) please go to http://www.bmj.com/company/products-services/rights-and-licensing/
Figures

References
-
- Strand V, Fiorentino D, Hu C, et al. . Improvements in patient-reported outcomes with apremilast, an oral phosphodiesterase 4 inhibitor, in the treatment of moderate to severe psoriasis: results from a phase IIb randomized, controlled study. Health Qual Life Outcomes 2013;11:82 10.1186/1477-7525-11-82 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

