A Temporal Switch in the Germinal Center Determines Differential Output of Memory B and Plasma Cells
- PMID: 26795247
- PMCID: PMC4724390
- DOI: 10.1016/j.immuni.2015.12.004
A Temporal Switch in the Germinal Center Determines Differential Output of Memory B and Plasma Cells
Abstract
There is little insight into or agreement about the signals that control differentiation of memory B cells (MBCs) and long-lived plasma cells (LLPCs). By performing BrdU pulse-labeling studies, we found that MBC formation preceded the formation of LLPCs in an adoptive transfer immunization system, which allowed for a synchronized Ag-specific response with homogeneous Ag-receptor, yet at natural precursor frequencies. We confirmed these observations in wild-type (WT) mice and extended them with germinal center (GC) disruption experiments and variable region gene sequencing. We thus show that the GC response undergoes a temporal switch in its output as it matures, revealing that the reaction engenders both MBC subsets with different immune effector function and, ultimately, LLPCs at largely separate points in time. These data demonstrate the kinetics of the formation of the cells that provide stable humoral immunity and therefore have implications for autoimmunity, for vaccine development, and for understanding long-term pathogen resistance.
Copyright © 2016 Elsevier Inc. All rights reserved.
Figures

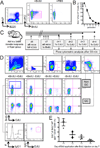


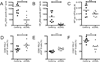

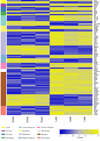
Comment in
-
Immune memory: Sequential evolution of B cell memory.Nat Rev Immunol. 2016 Feb;16(2):72-3. doi: 10.1038/nri.2016.15. Epub 2016 Jan 25. Nat Rev Immunol. 2016. PMID: 26806486 No abstract available.
References
-
- Allen CDC, Okada T, Tang HL, Cyster JG. Imaging of germinal center selection events during affinity maturation. Science. 2007b;315:528–531. - PubMed
-
- Anderson SM, Hannum LG, Shlomchik MJ. Memory B cell survival and function in the absence of secreted antibody and immune complexes on follicular dendritic cells. The Journal of Immunology. 2006;176:4515–4519. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

