Mechanisms Restricting Diffusion of Intracellular cAMP
- PMID: 26795432
- PMCID: PMC4726171
- DOI: 10.1038/srep19577
Mechanisms Restricting Diffusion of Intracellular cAMP
Abstract
Although numerous receptors stimulate cAMP production in a wide array of cells, many elicit distinct, highly localized responses, implying that the subcellular distribution of cAMP is not uniform. One often used explanation is that phosphodiesterases, which breakdown cAMP, act as functional barriers limiting diffusion. However, several studies refute the notion that this is sufficient, suggesting that phosphodiesterase-independent movement of cAMP must occur at rates slower than free diffusion. But, until now this has never been demonstrated. Using Raster Image Correlation Spectroscopy (RICS), we measured the diffusion coefficient of a fluorescently-labeled cAMP derivative (φ450-cAMP) as well as other fluorescent molecules in order to investigate the role that molecular size, cell morphology, and buffering by protein kinase A (PKA) play in restricting cAMP mobility in different cell types. Our results demonstrate that cytosolic movement of cAMP is indeed much slower than the rate of free diffusion and that interactions with PKA, especially type II PKA associated with mitochondria, play a significant role. These findings have important implications with respect to cAMP signaling in all cells.
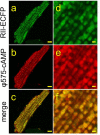
Figures





References
-
- Brunton L. L., Hayes J. S. & Mayer S. E. Functional compartmentation of cyclic AMP and protein kinase in heart. Advances in cyclic nucleotide research 14, 391–397 (1981). - PubMed
-
- Steinberg S. F. & Brunton L. L. Compartmentation of G protein-coupled signaling pathways in cardiac myocytes. Annu. Rev. Pharmacol. Toxicol. 41, 751–773 (2001). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

