A platform for brain-wide imaging and reconstruction of individual neurons
- PMID: 26796534
- PMCID: PMC4739768
- DOI: 10.7554/eLife.10566
A platform for brain-wide imaging and reconstruction of individual neurons
Abstract
The structure of axonal arbors controls how signals from individual neurons are routed within the mammalian brain. However, the arbors of very few long-range projection neurons have been reconstructed in their entirety, as axons with diameters as small as 100 nm arborize in target regions dispersed over many millimeters of tissue. We introduce a platform for high-resolution, three-dimensional fluorescence imaging of complete tissue volumes that enables the visualization and reconstruction of long-range axonal arbors. This platform relies on a high-speed two-photon microscope integrated with a tissue vibratome and a suite of computational tools for large-scale image data. We demonstrate the power of this approach by reconstructing the axonal arbors of multiple neurons in the motor cortex across a single mouse brain.
Keywords: Axonal reconstruction; Neuroanatomy; Neuroinformatics; Tissue clearing; Whole-brain imaging; mouse; neuroscience.
Conflict of interest statement
The author declares that no competing interests exist.
Figures
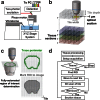

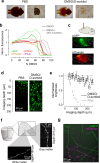

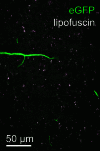






Comment in
-
Whole-brain imaging reaches new heights (and lengths).Elife. 2016 Jan 20;5:e13367. doi: 10.7554/eLife.13367. Elife. 2016. PMID: 26789654 Free PMC article.
References
-
- Bohland JW, Wu C, Barbas H, Bokil H, Bota M, Breiter HC, Cline HT, Doyle JC, Freed PJ, Greenspan RJ, Haber SN, Hawrylycz M, Herrera DG, Hilgetag CC, Huang ZJ, Jones A, Jones EG, Karten HJ, Kleinfeld D, Kötter R, Lester HA, Lin JM, Mensh BD, Mikula S, Panksepp J, Price JL, Safdieh J, Saper CB, Schiff ND, Schmahmann JD, Stillman BW, Svoboda K, Swanson LW, Toga AW, Van Essen DC, Watson JD, Mitra PP. A proposal for a coordinated effort for the determination of brainwide neuroanatomical connectivity in model organisms at a mesoscopic scale. PLoS Computational Biology. 2009;5:e10566. doi: 10.1371/journal.pcbi.1000334. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

