t-BHQ Provides Protection against Lead Neurotoxicity via Nrf2/HO-1 Pathway
- PMID: 26798413
- PMCID: PMC4698940
- DOI: 10.1155/2016/2075915
t-BHQ Provides Protection against Lead Neurotoxicity via Nrf2/HO-1 Pathway
Abstract
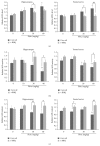
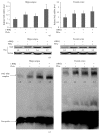
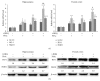
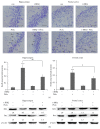
The neurotoxicity of lead has been well established, and oxidative stress is strongly associated with lead-induced neurotoxicity. Nrf2 is important for protection against oxidative stress in many disease models. We applied t-BHQ, which is an Nrf2 activator, to investigate the possible role of Nrf2 in the protection against lead neurotoxicity. t-BHQ significantly attenuated the oxidative stress in developmental rats by decreasing MDA level, as well as by increasing SOD activity and GSH content, in the hippocampus and frontal cortex. Furthermore, neuronal apoptosis was detected by Nissl staining, and Bax expression was inhibited in the t-BHQ-treated group. Results showed that t-BHQ suppressed ROS production and caspase 3/7 activity but increased intracellular GSH content, in SH-SY5Y cells under lead exposure. Moreover, in vivo and in vitro, t-BHQ enhanced the nuclear translocation of Nrf2 and binding to ARE areas but did not induce Nrf2 transcription. These phenomena were confirmed using RT-PCR, EMSA, Western blot, and immunofluorescence analyses. Subsequent upregulation of the expression of HO-1, NQO1, and GCLC was observed. However, knockdown of Nrf2 or HO-1 adversely affected the protective effects of t-BHQ against lead toxicity in SH-SY5Y cells. Thus, t-BHQ can protect against lead neurotoxicity, depending on the Nrf2/HO-1 pathway.
Figures


Similar articles
-
Neuroprotection against 6-OHDA-induced oxidative stress and apoptosis in SH-SY5Y cells by 5,7-Dihydroxychromone: Activation of the Nrf2/ARE pathway.Life Sci. 2015 Jun 1;130:25-30. doi: 10.1016/j.lfs.2015.02.026. Epub 2015 Mar 26. Life Sci. 2015. PMID: 25818191
-
Inhibition of beta-amyloid-induced neurotoxicity by pinocembrin through Nrf2/HO-1 pathway in SH-SY5Y cells.J Neurol Sci. 2016 Sep 15;368:223-30. doi: 10.1016/j.jns.2016.07.010. Epub 2016 Jul 11. J Neurol Sci. 2016. PMID: 27538638
-
Tert-butylhydroquinone attenuates the ethanol-induced apoptosis of and activates the Nrf2 antioxidant defense pathway in H9c2 cardiomyocytes.Int J Mol Med. 2016 Jul;38(1):123-30. doi: 10.3892/ijmm.2016.2605. Epub 2016 May 25. Int J Mol Med. 2016. PMID: 27220726 Free PMC article.
-
Efficiency of Traditional Chinese medicine targeting the Nrf2/HO-1 signaling pathway.Biomed Pharmacother. 2020 Jun;126:110074. doi: 10.1016/j.biopha.2020.110074. Epub 2020 Mar 9. Biomed Pharmacother. 2020. PMID: 32163746 Review.
-
Involvement of Nrf2 Signaling in Lead-induced Toxicity.Curr Med Chem. 2024;31(23):3529-3549. doi: 10.2174/0929867330666230522143341. Curr Med Chem. 2024. PMID: 37221680 Review.
Cited by
-
Identification and Validation of Endoplasmic Reticulum Stress-Related Gene in Traumatic Brain Injury.J Mol Neurosci. 2024 Sep 12;74(3):87. doi: 10.1007/s12031-024-02265-9. J Mol Neurosci. 2024. PMID: 39264510
-
Myricitrin Attenuates High Glucose-Induced Apoptosis through Activating Akt-Nrf2 Signaling in H9c2 Cardiomyocytes.Molecules. 2016 Jul 5;21(7):880. doi: 10.3390/molecules21070880. Molecules. 2016. PMID: 27399653 Free PMC article.
-
Molecular mechanisms and therapeutic effects of different vitamins and minerals in COVID-19 patients.J Trace Elem Med Biol. 2022 Sep;73:127044. doi: 10.1016/j.jtemb.2022.127044. Epub 2022 Jul 20. J Trace Elem Med Biol. 2022. PMID: 35901669 Free PMC article. Review.
-
A Novel STAT3-Mediated GATA6 Pathway Contributes to tert-Butylhydroquinone- (tBHQ-) Protected TNFα-Activated Vascular Cell Adhesion Molecule 1 (VCAM-1) in Vascular Endothelium.Oxid Med Cell Longev. 2020 Nov 14;2020:6584059. doi: 10.1155/2020/6584059. eCollection 2020. Oxid Med Cell Longev. 2020. PMID: 33274004 Free PMC article.
-
Iron homeostasis and post-hemorrhagic hydrocephalus: a review.Front Neurol. 2024 Jan 12;14:1287559. doi: 10.3389/fneur.2023.1287559. eCollection 2023. Front Neurol. 2024. PMID: 38283681 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

