Myocyte repolarization modulates myocardial function in aging dogs
- PMID: 26801307
- PMCID: PMC4867360
- DOI: 10.1152/ajpheart.00682.2015
Myocyte repolarization modulates myocardial function in aging dogs
Abstract
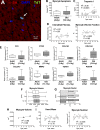
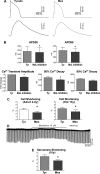
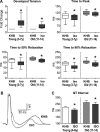
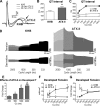
Studies of myocardial aging are complex and the mechanisms involved in the deterioration of ventricular performance and decreased functional reserve of the old heart remain to be properly defined. We have studied a colony of beagle dogs from 3 to 14 yr of age kept under a highly regulated environment to define the effects of aging on the myocardium. Ventricular, myocardial, and myocyte function, together with anatomical and structural properties of the organ and cardiomyocytes, were evaluated. Ventricular hypertrophy was not observed with aging and the structural composition of the myocardium was modestly affected. Alterations in the myocyte compartment were identified in aged dogs, and these factors negatively interfere with the contractile reserve typical of the young heart. The duration of the action potential is prolonged in old cardiomyocytes contributing to the slower electrical recovery of the myocardium. Also, the remodeled repolarization of cardiomyocytes with aging provides inotropic support to the senescent muscle but compromises its contractile reserve, rendering the old heart ineffective under conditions of high hemodynamic demand. The defects in the electrical and mechanical properties of cardiomyocytes with aging suggest that this cell population is an important determinant of the cardiac senescent phenotype. Collectively, the delayed electrical repolarization of aging cardiomyocytes may be viewed as a critical variable of the aging myopathy and its propensity to evolve into ventricular decompensation under stressful conditions.
Keywords: aging; contractile reserve; myocardium.
Copyright © 2016 the American Physiological Society.
Figures










Similar articles
-
The young mouse heart is composed of myocytes heterogeneous in age and function.Circ Res. 2007 Aug 17;101(4):387-99. doi: 10.1161/CIRCRESAHA.107.151449. Epub 2007 Jun 29. Circ Res. 2007. PMID: 17601802
-
The impact of age and frailty on ventricular structure and function in C57BL/6J mice.J Physiol. 2017 Jun 15;595(12):3721-3742. doi: 10.1113/JP274134. Epub 2017 May 14. J Physiol. 2017. PMID: 28502095 Free PMC article.
-
Late Na(+) current and protracted electrical recovery are critical determinants of the aging myopathy.Nat Commun. 2015 Nov 6;6:8803. doi: 10.1038/ncomms9803. Nat Commun. 2015. PMID: 26541940 Free PMC article.
-
In vitro models of the cardiac microenvironment to study myocyte and non-myocyte crosstalk: bioinspired approaches beyond the polystyrene dish.J Physiol. 2017 Jun 15;595(12):3891-3905. doi: 10.1113/JP273100. Epub 2017 Feb 27. J Physiol. 2017. PMID: 28116799 Free PMC article. Review.
-
Cell-to-cell electrical interactions during early and late repolarization.J Cardiovasc Electrophysiol. 2006 May;17 Suppl 1:S8-S14. doi: 10.1111/j.1540-8167.2006.00379.x. J Cardiovasc Electrophysiol. 2006. PMID: 16686687 Review.
Cited by
-
Rhythm dynamics of the aging heart: an experimental study using conscious, restrained mice.Am J Physiol Heart Circ Physiol. 2020 Oct 1;319(4):H893-H905. doi: 10.1152/ajpheart.00379.2020. Epub 2020 Sep 4. Am J Physiol Heart Circ Physiol. 2020. PMID: 32886003 Free PMC article.
-
Hyperglycemia induces defective Ca2+ homeostasis in cardiomyocytes.Am J Physiol Heart Circ Physiol. 2017 Jan 1;312(1):H150-H161. doi: 10.1152/ajpheart.00737.2016. Epub 2016 Nov 23. Am J Physiol Heart Circ Physiol. 2017. PMID: 27881388 Free PMC article.
-
Depleted Myocardial Coenzyme Q10 in Cavalier King Charles Spaniels with Congestive Heart Failure Due to Myxomatous Mitral Valve Disease.Antioxidants (Basel). 2021 Jan 22;10(2):161. doi: 10.3390/antiox10020161. Antioxidants (Basel). 2021. PMID: 33499156 Free PMC article.
-
Phosphorylation of cardiac sodium channel at Ser571 anticipates manifestations of the aging myopathy.Am J Physiol Heart Circ Physiol. 2024 Jun 1;326(6):H1424-H1445. doi: 10.1152/ajpheart.00325.2023. Epub 2024 Apr 19. Am J Physiol Heart Circ Physiol. 2024. PMID: 38639742 Free PMC article.
-
Involvement of TGF-β, mTOR, and inflammatory mediators in aging alterations during myxomatous mitral valve disease in a canine model.Geroscience. 2025 Jan 27. doi: 10.1007/s11357-025-01520-0. Online ahead of print. Geroscience. 2025. PMID: 39865135
References
-
- Belardinelli L, Giles WR, Rajamani S, Karagueuzian HS, Shryock JC. Cardiac late Na(+) current: proarrhythmic effects, roles in long QT syndromes, and pathological relationship to CaMKII and oxidative stress. Heart Rhythm 12: 440–448, 2015. - PubMed
-
- Bernstein RD, Zhang X, Zhao G, Forfia P, Tuzman J, Ochoa F, Ochoa M, Vogel T, Hintze TH. Mechanisms of nitrate accumulation in plasma during pacing-induced heart failure in conscious dogs. Nitric Oxide 1: 386–396, 1997. - PubMed
-
- Boyle AJ, Shih H, Hwang J, Ye J, Lee B, Zhang Y, Kwon D, Jun K, Zheng D, Sievers R, Angeli F, Yeghiazarians Y, Lee R. Cardiomyopathy of aging in the mammalian heart is characterized by myocardial hypertrophy, fibrosis and a predisposition towards cardiomyocyte apoptosis and autophagy. Exp Gerontol 46: 549–559, 2011. - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

