MicroRNA-7 Regulates the Function of Mitochondrial Permeability Transition Pore by Targeting VDAC1 Expression
- PMID: 26801612
- PMCID: PMC4813563
- DOI: 10.1074/jbc.M115.691352
MicroRNA-7 Regulates the Function of Mitochondrial Permeability Transition Pore by Targeting VDAC1 Expression
Abstract
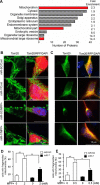
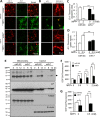
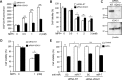
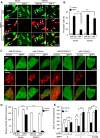
Mitochondrial dysfunction is one of the major contributors to neurodegenerative disorders including Parkinson disease. The mitochondrial permeability transition pore is a protein complex located on the mitochondrial membrane. Under cellular stress, the pore opens, increasing the release of pro-apoptotic proteins, and ultimately resulting in cell death. MicroRNA-7 (miR-7) is a small non-coding RNA that has been found to exhibit a protective role in the cellular models of Parkinson disease. In the present study, miR-7 was predicted to regulate the function of mitochondria, according to gene ontology analysis of proteins that are down-regulated by miR-7. Indeed, miR-7 overexpression inhibited mitochondrial fragmentation, mitochondrial depolarization, cytochrome c release, reactive oxygen species generation, and release of mitochondrial calcium in response to 1-methyl-4-phenylpyridinium (MPP(+)) in human neuroblastoma SH-SY5Y cells. In addition, several of these findings were confirmed in mouse primary neurons. Among the mitochondrial proteins identified by gene ontology analysis, the expression of voltage-dependent anion channel 1 (VDAC1), a constituent of the mitochondrial permeability transition pore, was down-regulated by miR-7 through targeting 3'-untranslated region of VDAC1 mRNA. Similar to miR-7 overexpression, knockdown of VDAC1 also led to a decrease in intracellular reactive oxygen species generation and subsequent cellular protection against MPP(+). Notably, overexpression of VDAC1 without the 3'-UTR significantly abolished the protective effects of miR-7 against MPP(+)-induced cytotoxicity and mitochondrial dysfunction, suggesting that the protective effect of miR-7 is partly exerted through promoting mitochondrial function by targeting VDAC1 expression. These findings point to a novel mechanism by which miR-7 accomplishes neuroprotection by improving mitochondrial health.
Keywords: 1-methyl-4-phenylpyridinium; Parkinson disease; microRNA (miRNA); mitochondrial membrane potential; mitochondrial permeability transition (MPT); voltage-dependent anion channel (VDAC).
© 2016 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures

Similar articles
-
Voltage-dependent anion channels are dispensable for mitochondrial-dependent cell death.Nat Cell Biol. 2007 May;9(5):550-5. doi: 10.1038/ncb1575. Epub 2007 Apr 8. Nat Cell Biol. 2007. PMID: 17417626 Free PMC article.
-
Outer membrane VDAC1 controls permeability transition of the inner mitochondrial membrane in cellulo during stress-induced apoptosis.Cell Res. 2009 Dec;19(12):1363-76. doi: 10.1038/cr.2009.98. Epub 2009 Aug 11. Cell Res. 2009. PMID: 19668262
-
The MicroRNA-224 Inhibitor Prevents Neuronal Apoptosis via Targeting Spastic Paraplegia 7 After Cerebral Ischemia.J Mol Neurosci. 2016 Jul;59(3):421-9. doi: 10.1007/s12031-016-0769-9. Epub 2016 May 10. J Mol Neurosci. 2016. PMID: 27165196
-
The mitochondrial voltage-dependent anion channel 1 in tumor cells.Biochim Biophys Acta. 2015 Oct;1848(10 Pt B):2547-75. doi: 10.1016/j.bbamem.2014.10.040. Epub 2014 Nov 4. Biochim Biophys Acta. 2015. PMID: 25448878 Review.
-
Targeting putative components of the mitochondrial permeability transition pore for novel therapeutics.Biochem Pharmacol. 2020 Jul;177:113995. doi: 10.1016/j.bcp.2020.113995. Epub 2020 Apr 25. Biochem Pharmacol. 2020. PMID: 32339494 Review.
Cited by
-
Various Aspects of Calcium Signaling in the Regulation of Apoptosis, Autophagy, Cell Proliferation, and Cancer.Int J Mol Sci. 2020 Nov 6;21(21):8323. doi: 10.3390/ijms21218323. Int J Mol Sci. 2020. PMID: 33171939 Free PMC article. Review.
-
Decoding Cancer through Silencing the Mitochondrial Gatekeeper VDAC1.Biomolecules. 2024 Oct 15;14(10):1304. doi: 10.3390/biom14101304. Biomolecules. 2024. PMID: 39456237 Free PMC article. Review.
-
The mitomiR/Bcl-2 axis affects mitochondrial function and autophagic vacuole formation in senescent endothelial cells.Aging (Albany NY). 2018 Oct 21;10(10):2855-2873. doi: 10.18632/aging.101591. Aging (Albany NY). 2018. PMID: 30348904 Free PMC article.
-
Mitochondrial Dysfunction and Parkinson's Disease: Pathogenesis and Therapeutic Strategies.Neurochem Res. 2023 Aug;48(8):2285-2308. doi: 10.1007/s11064-023-03904-0. Epub 2023 Mar 21. Neurochem Res. 2023. PMID: 36943668 Review.
-
Epigenetic mechanisms of neurodegenerative diseases and acute brain injury.Neurochem Int. 2020 Feb;133:104642. doi: 10.1016/j.neuint.2019.104642. Epub 2019 Dec 12. Neurochem Int. 2020. PMID: 31838024 Free PMC article. Review.
References
-
- Abou-Sleiman P. M., Muqit M. M., and Wood N. W. (2006) Expanding insights of mitochondrial dysfunction in Parkinson's disease. Nat. Rev. Neurosci. 7, 207–219 - PubMed
-
- Duchen M. R. (2004) Mitochondria in health and disease: perspectives on a new mitochondrial biology. Mol. Aspects Med. 25, 365–451 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

