Effects of the pattern of glucocorticoid replacement on neural processing, emotional reactivity and well-being in healthy male individuals: study protocol for a randomised controlled trial
- PMID: 26801980
- PMCID: PMC4724084
- DOI: 10.1186/s13063-016-1159-x
Effects of the pattern of glucocorticoid replacement on neural processing, emotional reactivity and well-being in healthy male individuals: study protocol for a randomised controlled trial
Abstract
Background: Deviation from the physiological glucocorticoid dynamics (circadian and underlying ultradian rhythmicity) is a common characteristic of various neuropsychiatric and endocrine disorders as well as glucocorticoid-based therapeutics. These states may be accompanied by neuropsychiatric symptomatology, suggesting continuous dynamic glucocorticoid equilibrium is essential for brain homeostasis.
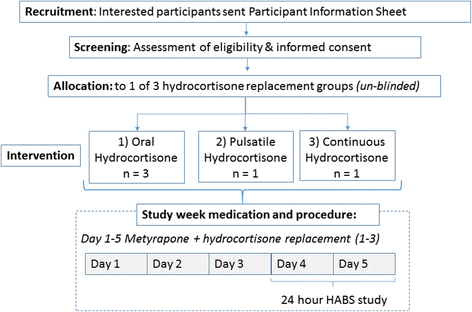
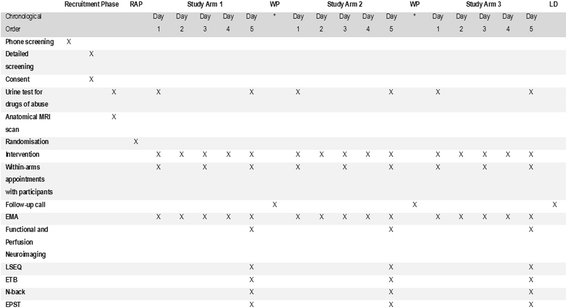
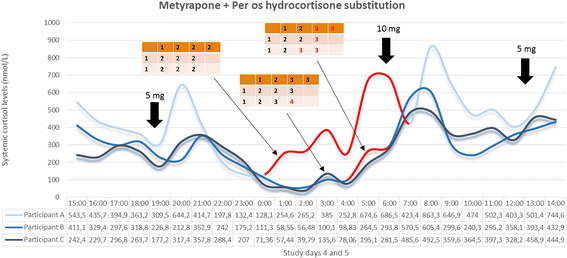
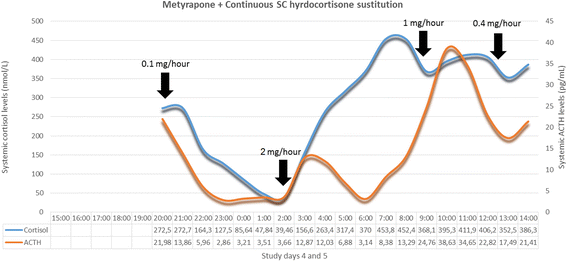
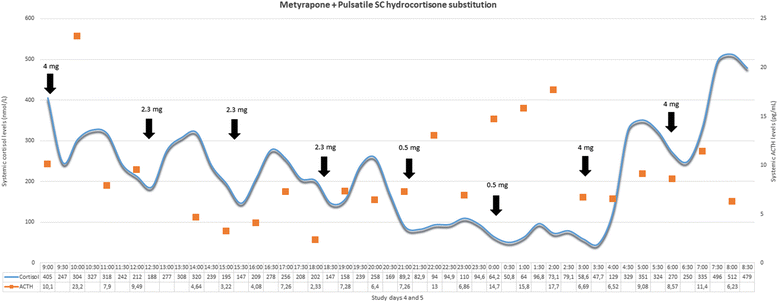
Methods/design: The study consists of two parts. The preliminary stage of the study aims to validate (technically and pharmacologically) and optimise three different patterns of systemic cortisol administration in man. These patterns are based on the combinatory administration of metyrapone, to suppress endogenous cortisol production, and concurrent hydrocortisone replacement. The second, subsequent, core part of the study is a randomised, double-blinded, placebo-controlled, crossover study, where participants (healthy male individuals aged 18-60 years) will undergo all three hydrocortisone replacement schemes. During these infusion regimes, we plan a number of neurobehavioural tests and imaging of the brain to assess neural processing, emotional reactivity and perception, mood and self-perceived well-being. The psychological tests include: ecological momentary assessment, P1vital Oxford Emotional Test Battery and Emotional Potentiated Startle Test, Leeds Sleep Evaluation Questionnaire and the visual working memory task (n-back). The neuroimaging protocol combines magnetic resonance sequences that capture data related to the functional and perfusion status of the brain.
Discussion: Results of this clinical trial are designed to evaluate the impact (with possible mechanistic insights) of different patterns of daily glucocorticoid dynamics on neural processing and reactivity related to emotional perception and mood. This evidence should contribute to the optimisation of the clinical application of glucocorticoid-based therapeutics.
Trial registration: UK Clinical Research Network, IRAS Ref: 106181, UKCRN-ID-15236 (23 October 2013).
Figures

Similar articles
-
Glucocorticoid ultradian rhythmicity differentially regulates mood and resting state networks in the human brain: A randomised controlled clinical trial.Psychoneuroendocrinology. 2021 Feb;124:105096. doi: 10.1016/j.psyneuen.2020.105096. Epub 2020 Dec 1. Psychoneuroendocrinology. 2021. PMID: 33296841 Free PMC article. Clinical Trial.
-
Ultradian hydrocortisone replacement alters neuronal processing, emotional ambiguity, affect and fatigue in adrenal insufficiency: The PULSES trial.J Intern Med. 2024 Jan;295(1):51-67. doi: 10.1111/joim.13721. Epub 2023 Oct 19. J Intern Med. 2024. PMID: 37857352 Free PMC article. Clinical Trial.
-
Prednisolone increases neural reactivity to negative socio-emotional stimuli in healthy young men.Eur Neuropsychopharmacol. 2016 Jul;26(7):1176-89. doi: 10.1016/j.euroneuro.2016.04.010. Epub 2016 May 11. Eur Neuropsychopharmacol. 2016. PMID: 27178366 Clinical Trial.
-
Folic acid supplementation and malaria susceptibility and severity among people taking antifolate antimalarial drugs in endemic areas.Cochrane Database Syst Rev. 2022 Feb 1;2(2022):CD014217. doi: 10.1002/14651858.CD014217. Cochrane Database Syst Rev. 2022. PMID: 36321557 Free PMC article.
-
Glucocorticoid replacement regimens for treating congenital adrenal hyperplasia.Cochrane Database Syst Rev. 2020 Mar 19;3(3):CD012517. doi: 10.1002/14651858.CD012517.pub2. Cochrane Database Syst Rev. 2020. PMID: 32190901 Free PMC article.
Cited by
-
Mineralocorticoid receptor and glucocorticoid receptor work alone and together in cell-type-specific manner: Implications for resilience prediction and targeted therapy.Neurobiol Stress. 2022 Apr 22;18:100455. doi: 10.1016/j.ynstr.2022.100455. eCollection 2022 May. Neurobiol Stress. 2022. PMID: 35601687 Free PMC article.
-
Glucocorticoid ultradian rhythmicity differentially regulates mood and resting state networks in the human brain: A randomised controlled clinical trial.Psychoneuroendocrinology. 2021 Feb;124:105096. doi: 10.1016/j.psyneuen.2020.105096. Epub 2020 Dec 1. Psychoneuroendocrinology. 2021. PMID: 33296841 Free PMC article. Clinical Trial.
-
Modelling Hydrocortisone Pharmacokinetics on a Subcutaneous Pulsatile Infusion Replacement Strategy in Patients with Adrenocortical Insufficiency.Pharmaceutics. 2021 May 21;13(6):769. doi: 10.3390/pharmaceutics13060769. Pharmaceutics. 2021. PMID: 34064165 Free PMC article.
-
Transcriptional and cell type profiles of cortical brain regions showing ultradian cortisol rhythm dependent responses to emotional face stimulation.Neurobiol Stress. 2023 Jan 4;22:100514. doi: 10.1016/j.ynstr.2023.100514. eCollection 2023 Jan. Neurobiol Stress. 2023. PMID: 36660181 Free PMC article.
-
Ultradian hydrocortisone replacement alters neuronal processing, emotional ambiguity, affect and fatigue in adrenal insufficiency: The PULSES trial.J Intern Med. 2024 Jan;295(1):51-67. doi: 10.1111/joim.13721. Epub 2023 Oct 19. J Intern Med. 2024. PMID: 37857352 Free PMC article. Clinical Trial.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

