Genetic and pharmacological evidence that endogenous nociceptin/orphanin FQ contributes to dopamine cell loss in Parkinson's disease
- PMID: 26804029
- PMCID: PMC5567676
- DOI: 10.1016/j.nbd.2016.01.016
Genetic and pharmacological evidence that endogenous nociceptin/orphanin FQ contributes to dopamine cell loss in Parkinson's disease
Abstract
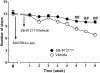
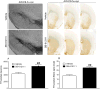
To investigate whether the endogenous neuropeptide nociceptin/orphanin FQ (N/OFQ) contributes to the death of dopamine neurons in Parkinson's disease, we undertook a genetic and a pharmacological approach using NOP receptor knockout (NOP(-/-)) mice, and the selective and potent small molecule NOP receptor antagonist (-)-cis-1-methyl-7-[[4-(2,6-dichlorophenyl)piperidin-1-yl]methyl]-6,7,8,9-tetrahydro-5H-benzocyclohepten-5-ol (SB-612111). Stereological unbiased methods were used to estimate the total number of dopamine neurons in the substantia nigra of i) NOP(-/-) mice acutely treated with the parkinsonian neurotoxin 1-methyl-4-phenyl-1,2,5,6-tetrahydropyridine (MPTP), ii) naïve mice subacutely treated with MPTP, alone or in combination with SB-612111, iii) rats injected with a recombinant adeno-associated viral (AAV) vector overexpressing human mutant p.A53T α-synuclein, treated with vehicle or SB-612111. NOP(-/-) mice showed a 50% greater amount of nigral dopamine neurons spared in response to acute MPTP compared to controls, which was associated with a milder motor impairment. SB-612111, given 4 days after MPTP treatment to mimic the clinical condition, prevented the loss of nigral dopamine neurons and striatal dopaminergic terminals caused by subacute MPTP. SB-612111, administered a week after the AAV injections in a clinically-driven protocol, also increased by 50% both the number of spared nigral dopamine neurons and striatal dopamine terminals, and prevented accompanying motor deficits induced by α-synuclein. We conclude that endogenous N/OFQ contributes to dopamine neuron loss in pathogenic and etiologic models of Parkinson's disease through NOP receptor-mediated mechanisms. NOP receptor antagonists might prove effective as disease-modifying agents in Parkinson's disease, through the rescue of degenerating nigral dopamine neurons and/or the protection of the healthy ones.
Keywords: Adeno associated viral vectors; MPP(+); MPTP; NOP receptor; Neuroprotection; Nociceptin/orphanin FQ; Parkinson's disease; SB-612111; α-Synuclein.
Copyright © 2016. Published by Elsevier Inc.
Figures




References
-
- Bezard E. Neuroprotection for Parkinson's disease: a call for clinically driven experimental design. Lancet Neurol. 2003;2:393. - PubMed
-
- Brown JM, et al. Differential protection against MPTP or methamphetamine toxicity in dopamine neurons by deletion of ppN/OFQ expression. J. Neurochem. 2006;98:495–505. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

