A Simple Hydraulic Analog Model of Oxidative Phosphorylation
- PMID: 26807634
- PMCID: PMC6293281
- DOI: 10.1249/MSS.0000000000000884
A Simple Hydraulic Analog Model of Oxidative Phosphorylation
Abstract
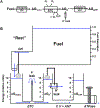
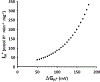
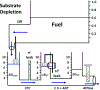
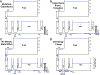
Mitochondrial oxidative phosphorylation is the primary source of cellular energy transduction in mammals. This energy conversion involves dozens of enzymatic reactions, energetic intermediates, and the dynamic interactions among them. With the goal of providing greater insight into the complex thermodynamics and kinetics ("thermokinetics") of mitochondrial energy transduction, a simple hydraulic analog model of oxidative phosphorylation is presented. In the hydraulic model, water tanks represent the forward and back "pressures" exerted by thermodynamic driving forces: the matrix redox potential (ΔGredox), the electrochemical potential for protons across the mitochondrial inner membrane (ΔGH), and the free energy of adenosine 5'-triphosphate (ATP) (ΔGATP). Net water flow proceeds from tanks with higher water pressure to tanks with lower pressure through "enzyme pipes" whose diameters represent the conductances (effective activities) of the proteins that catalyze the energy transfer. These enzyme pipes include the reactions of dehydrogenase enzymes, the electron transport chain (ETC), and the combined action of ATP synthase plus the ATP-adenosine 5'-diphosphate exchanger that spans the inner membrane. In addition, reactive oxygen species production is included in the model as a leak that is driven out of the ETC pipe by high pressure (high ΔGredox) and a proton leak dependent on the ΔGH for both its driving force and the conductance of the leak pathway. Model water pressures and flows are shown to simulate thermodynamic forces and metabolic fluxes that have been experimentally observed in mammalian skeletal muscle in response to acute exercise, chronic endurance training, and reduced substrate availability, as well as account for the thermokinetic behavior of mitochondria from fast- and slow-twitch skeletal muscle and the metabolic capacitance of the creatine kinase reaction.
Figures


References
-
- Ackrell BA, Maguire JJ, Dallman PR, Kearney EB. Effect of iron deficiency on succinate-and NADH-ubiquinone oxidoreductases in skeletal muscle mitochondria. J Biol Chem 1984;259(16):10053–9. - PubMed
-
- Balaban RS, Kantor HL, Katz LA, Briggs RW. Relation between work and phosphate metabolite in the in vivo paced mammalian heart. Science 1986;232(4754):1121–3. - PubMed
-
- Baldwin KM, Fitts RH, Booth FW, Winder WW, Holloszy JO. Depletion of muscle and liver glycogen during exercise. Protective effect of training. Pflugers Arch 1975;354(3):203–12. - PubMed
-
- Brand MD. The proton leak across the mitochondrial inner membrane. Biochim Biophys Acta 1990;1018(2–3):128–33. - PubMed
-
- Brand MD. Top down metabolic control analysis. J Theor Biol 1996;182(3):351–60. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

