Ca2+/Calmodulin-Dependent Protein Kinase Kinases (CaMKKs) Effects on AMP-Activated Protein Kinase (AMPK) Regulation of Chicken Sperm Functions
- PMID: 26808520
- PMCID: PMC4726612
- DOI: 10.1371/journal.pone.0147559
Ca2+/Calmodulin-Dependent Protein Kinase Kinases (CaMKKs) Effects on AMP-Activated Protein Kinase (AMPK) Regulation of Chicken Sperm Functions
Abstract
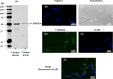
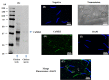
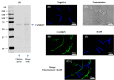
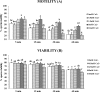
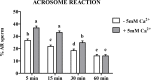
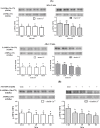
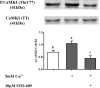
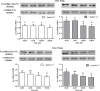
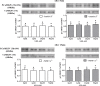
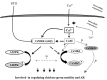
Sperm require high levels of energy to ensure motility and acrosome reaction (AR) accomplishment. The AMP-activated protein kinase (AMPK) has been demonstrated to be strongly involved in the control of these properties. We address here the question of the potential role of calcium mobilization on AMPK activation and function in chicken sperm through the Ca(2+)/calmodulin-dependent protein kinase kinases (CaMKKs) mediated pathway. The presence of CaMKKs and their substrates CaMKI and CaMKIV was evaluated by western-blotting and indirect immunofluorescence. Sperm were incubated in presence or absence of extracellular Ca(2+), or of CaMKKs inhibitor (STO-609). Phosphorylations of AMPK, CaMKI, and CaMKIV, as well as sperm functions were evaluated. We demonstrate the presence of both CaMKKs (α and β), CaMKI and CaMKIV in chicken sperm. CaMKKα and CaMKI were localized in the acrosome, the midpiece, and at much lower fluorescence in the flagellum, whereas CaMKKβ was mostly localized in the flagellum and much less in the midpiece and the acrosome. CaMKIV was only present in the flagellum. The presence of extracellular calcium induced an increase in kinases phosphorylation and sperm activity. STO-609 reduced AMPK phosphorylation in the presence of extracellular Ca(2+) but not in its absence. STO-609 did not affect CaMKIV phosphorylation but decreased CaMKI phosphorylation and this inhibition was quicker in the presence of extracellular Ca(2+) than in its absence. STO-609 efficiently inhibited sperm motility and AR, both in the presence and absence of extracellular Ca(2+). Our results show for the first time the presence of CaMKKs (α and β) and one of its substrate, CaMKI in different subcellular compartments in germ cells, as well as the changes in the AMPK regulation pathway, sperm motility and AR related to Ca(2+) entry in sperm through the Ca(2+)/CaM/CaMKKs/CaMKI pathway. The Ca(2+)/CaMKKs/AMPK pathway is activated only under conditions of extracellular Ca(2+) entry in the cells.
Conflict of interest statement
Figures


Similar articles
-
Differential AMP-activated Protein Kinase (AMPK) Recognition Mechanism of Ca2+/Calmodulin-dependent Protein Kinase Kinase Isoforms.J Biol Chem. 2016 Jun 24;291(26):13802-8. doi: 10.1074/jbc.M116.727867. Epub 2016 May 5. J Biol Chem. 2016. PMID: 27151216 Free PMC article.
-
The calcium/CaMKKalpha/beta and the cAMP/PKA pathways are essential upstream regulators of AMPK activity in boar spermatozoa.Biol Reprod. 2014 Feb 13;90(2):29. doi: 10.1095/biolreprod.113.112797. Print 2014 Feb. Biol Reprod. 2014. PMID: 24389872
-
Possible CaMKK-dependent regulation of AMPK phosphorylation and glucose uptake at the onset of mild tetanic skeletal muscle contraction.Am J Physiol Endocrinol Metab. 2007 May;292(5):E1308-17. doi: 10.1152/ajpendo.00456.2006. Epub 2007 Jan 9. Am J Physiol Endocrinol Metab. 2007. PMID: 17213473
-
Molecular Mechanisms Underlying Ca2+/Calmodulin-Dependent Protein Kinase Kinase Signal Transduction.Int J Mol Sci. 2022 Sep 20;23(19):11025. doi: 10.3390/ijms231911025. Int J Mol Sci. 2022. PMID: 36232320 Free PMC article. Review.
-
New insights into transduction pathways that regulate boar sperm function.Theriogenology. 2016 Jan 1;85(1):12-20. doi: 10.1016/j.theriogenology.2015.05.008. Epub 2015 May 14. Theriogenology. 2016. PMID: 26074068 Review.
Cited by
-
The Seminal fluid proteome of the polyandrous Red junglefowl offers insights into the molecular basis of fertility, reproductive ageing and domestication.Sci Rep. 2016 Nov 2;6:35864. doi: 10.1038/srep35864. Sci Rep. 2016. PMID: 27804984 Free PMC article.
-
AMPK Function in Mammalian Spermatozoa.Int J Mol Sci. 2018 Oct 23;19(11):3293. doi: 10.3390/ijms19113293. Int J Mol Sci. 2018. PMID: 30360525 Free PMC article. Review.
-
Investigating the impact of paternal aging on murine sperm miRNA profiles and their potential link to autism spectrum disorder.Sci Rep. 2023 Dec 7;13(1):20608. doi: 10.1038/s41598-023-47878-z. Sci Rep. 2023. PMID: 38062235 Free PMC article.
-
Membrane Hormone Receptors and Their Signaling Pathways as Targets for Endocrine Disruptors.J Xenobiot. 2022 Mar 25;12(2):64-73. doi: 10.3390/jox12020007. J Xenobiot. 2022. PMID: 35466213 Free PMC article. Review.
-
Natural AMPK Activators in Cardiovascular Disease Prevention.Front Pharmacol. 2022 Jan 3;12:738420. doi: 10.3389/fphar.2021.738420. eCollection 2021. Front Pharmacol. 2022. PMID: 35046800 Free PMC article. Review.
References
-
- Woods A, Dickerson K, Heath R, Hong SP, Momcilovic M, Johnstone SR, et al. (2005) Ca2+/calmodulin-dependent protein kinase kinase-beta acts upstream of AMP-activated protein kinase in mammalian cells. Cell Metab 2: 21–33. - PubMed
-
- Hawley SA, Pan DA, Mustard KJ, Ross L, Bain J, Edelman AM, et al. (2005) Calmodulin-dependent protein kinase kinase-beta is an alternative upstream kinase for AMP-activated protein kinase. Cell Metab 2: 9–19. - PubMed
-
- Blesbois E (2012) Biological features of the avian male gamete and their application to biotechnology of conservation. Journal of Poultry Science, 49: 141–149.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

