Pharmacokinetics of Rifampin, Isoniazid, Pyrazinamide, and Ethambutol in Infants Dosed According to Revised WHO-Recommended Treatment Guidelines
- PMID: 26810651
- PMCID: PMC4808214
- DOI: 10.1128/AAC.02600-15
Pharmacokinetics of Rifampin, Isoniazid, Pyrazinamide, and Ethambutol in Infants Dosed According to Revised WHO-Recommended Treatment Guidelines
Abstract
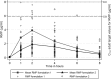
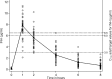
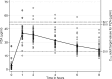
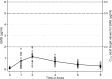
There are limited pharmacokinetic data for use of the first-line antituberculosis drugs during infancy (<12 months of age), when drug disposition may differ. Intensive pharmacokinetic sampling was performed in infants routinely receiving antituberculosis treatment, including rifampin, isoniazid, pyrazinamide, and ethambutol, using World Health Organization-recommended doses. Regulatory-approved single-drug formulations, including two rifampin suspensions, were used on the sampling day. Assays were conducted using liquid chromatography-mass spectrometry; pharmacokinetic parameters were generated using noncompartmental analysis. Thirty-nine infants were studied; 14 (36%) had culture-confirmed tuberculosis. Fifteen (38%) were premature (<37 weeks gestation); 5 (13%) were HIV infected. The mean corrected age and weight were 6.6 months and 6.45 kg, respectively. The mean maximum plasma concentrations (Cmax) for rifampin, isoniazid, pyrazinamide, and ethambutol were 2.9, 7.9, 41.9, and 1.3 μg/ml, respectively (current recommended adult target concentrations: 8 to 24, 3 to 6, 20 to 50, and 2 to 6 μg/ml, respectively), and the mean areas under the concentration-time curves from 0 to 8 h (AUC0-8) were 12.1, 24.7, 239.4, and 5.1 μg · h/ml, respectively. After adjusting for age and weight, rifampin exposures for the two formulations used differed inCmax(geometric mean ratio [GMR],2.55; 95% confidence interval [CI], 1.47 to 4.41;P= 0.001) and AUC0-8(GMR, 2.52; 95% CI, 1.34 to 4.73;P= 0.005). HIV status was associated with lower pyrazinamideCmax(GMR, 0.85; 95% CI, 0.75 to 0.96;P= 0.013) and AUC0-8(GMR, 0.79; 95% CI, 0.69 to 0.90;P< 0.001) values. No other important differences were observed due to age, weight, prematurity, ethnicity, or gender. In summary, isoniazid and pyrazinamide concentrations in infants compared well with proposed adult target concentrations; ethambutol concentrations were lower but similar to previously reported pediatric studies. The low rifampin exposures require further investigation. (This study has been registered at ClinicalTrials.gov under registration no. NCT01637558.).
Copyright © 2016, American Society for Microbiology. All Rights Reserved.
Figures
Similar articles
-
HIV-1 Coinfection Does Not Reduce Exposure to Rifampin, Isoniazid, and Pyrazinamide in South African Tuberculosis Outpatients.Antimicrob Agents Chemother. 2016 Sep 23;60(10):6050-9. doi: 10.1128/AAC.00480-16. Print 2016 Oct. Antimicrob Agents Chemother. 2016. PMID: 27480859 Free PMC article.
-
Pharmacokinetics of Antituberculosis Drugs in HIV-Positive and HIV-Negative Adults in Malawi.Antimicrob Agents Chemother. 2015 Oct;59(10):6175-80. doi: 10.1128/AAC.01193-15. Epub 2015 Jul 27. Antimicrob Agents Chemother. 2015. PMID: 26248378 Free PMC article. Clinical Trial.
-
Pharmacokinetics of the First-Line Antituberculosis Drugs in Ghanaian Children with Tuberculosis with or without HIV Coinfection.Antimicrob Agents Chemother. 2017 Jan 24;61(2):e01701-16. doi: 10.1128/AAC.01701-16. Print 2017 Feb. Antimicrob Agents Chemother. 2017. PMID: 27855070 Free PMC article. Clinical Trial.
-
Optimizing treatment outcome of first-line anti-tuberculosis drugs: the role of therapeutic drug monitoring.Eur J Clin Pharmacol. 2016 Aug;72(8):905-16. doi: 10.1007/s00228-016-2083-4. Epub 2016 Jun 15. Eur J Clin Pharmacol. 2016. PMID: 27305904 Review.
-
Chromatographic Methods for the Determination of Rifampicin, Isoniazid, Pyrazinamide, Ethambutol, and Main Metabolites in Biological Samples: A Review.Crit Rev Anal Chem. 2024;54(7):1971-1986. doi: 10.1080/10408347.2022.2150071. Epub 2022 Nov 29. Crit Rev Anal Chem. 2024. PMID: 36444713 Review.
Cited by
-
Pharmacokinetics of First-Line Drugs Among Children With Tuberculosis in Rural Tanzania.J Pediatric Infect Dis Soc. 2020 Feb 28;9(1):14-20. doi: 10.1093/jpids/piy106. J Pediatric Infect Dis Soc. 2020. PMID: 30395239 Free PMC article.
-
The Development of an Age-Appropriate Fixed Dose Combination for Tuberculosis Using Physiologically-Based Pharmacokinetic Modeling (PBBM) and Risk Assessment.Pharmaceutics. 2024 Dec 12;16(12):1587. doi: 10.3390/pharmaceutics16121587. Pharmaceutics. 2024. PMID: 39771565 Free PMC article.
-
A Systematic Review on the Effect of HIV Infection on the Pharmacokinetics of First-Line Tuberculosis Drugs.Clin Pharmacokinet. 2019 Jun;58(6):747-766. doi: 10.1007/s40262-018-0716-8. Clin Pharmacokinet. 2019. PMID: 30406475 Free PMC article.
-
Pharmacokinetics of First-Line Drugs in Children With Tuberculosis, Using World Health Organization-Recommended Weight Band Doses and Formulations.Clin Infect Dis. 2022 May 30;74(10):1767-1775. doi: 10.1093/cid/ciab725. Clin Infect Dis. 2022. PMID: 34420049 Free PMC article.
-
Revisiting the mutant prevention concentration to guide dosing in childhood tuberculosis.J Antimicrob Chemother. 2017 Jul 1;72(7):1848-1857. doi: 10.1093/jac/dkx051. J Antimicrob Chemother. 2017. PMID: 28333284 Free PMC article. Review.
References
-
- Marais BJ, Gie RP, Schaaf HS, Hesseling AC, Obihara CC, Starke JJ, Enarson DA, Donald PR, Beyers N. 2004. The natural history of childhood intrathoracic tuberculosis: a critical review of literature from the prechemotherapy era. Int J Tuberc Lung Dis 8:392–402. - PubMed
-
- Gupta A, Nayak U, Ram M, Bhosale R, Patil S, Basavraj A, Kakrani A, Philip S, Deasi D, Sastry J, Bollinger RC, Byramjee Jeejeebhoy Medical College-John Hopkins University Study Group. 2007. Postpartum tuberculosis incidence and mortality among HIV-infected women and their infants in Pune, India, 2002-2005. Clin Infect Dis 45:241–9. doi:10.1086/518974. - DOI - PubMed
-
- World Health Organization. 2010. Rapid advice: treatment of tuberculosis in children. WHO/HTM/TB/2010.13. World Health Organization, Geneva, Switzerland. - PubMed
-
- World Health Organization. 2006. Guidance for national tuberculosis programs on the management of tuberculosis in children. World Health Organization, Geneva, Switzerland.
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

