Sequence of treatment in locally advanced and metastatic renal cell carcinoma
- PMID: 26816832
- PMCID: PMC4708238
- DOI: 10.3978/j.issn.2223-4683.2015.04.07
Sequence of treatment in locally advanced and metastatic renal cell carcinoma
Abstract
The spectrum of drugs that have shown activity in advanced or metastatic renal cell carcinoma (RCC) has led to a debate on the optimal sequence of treatments. There is agreement on recommending targeted agents as the standard of care in this disease. Uncertainty, however, remains on the best first-line drug choice. Physicians and patients may select sunitinib, bevacizumab in combination with interferon-alpha (IFN-α), pazopanib, or-in poor risk patients-temsirolimus. There are also a variety of therapies with proven efficacy on hand in the second-line setting: sorafenib, pazopanib, axitinib, and everolimus. While most randomized RCC trials assessed progression free survival (PFS) as primary endpoint, some agents were shown to improve median overall survival (OS), and given in sequence they have extended the life expectancy of RCC patients from 13 months in the cytokine era to over 30 months. Despite the progress made, there are sobering aspects to the oncologic success story in RCC, as the new treatments do not obtain an objective response or disease stabilization (SD) in all patients. There are also as yet no predictors to select patients who might benefit and those who are primary resistant to specific drugs, and ultimately almost all patients will experience disease progression. Bearing inevitable treatment failure in mind, availability of further drugs and switching therapy while the patient is in a condition to continue pharmacotherapy is essential. Of note, depending on the setting, only 33-59% of patients receive second-line treatment. In this review we present data on first-, second-, and third-line treatment in RCC, and discuss the difficulties in their interpretation in the context of treatment sequence. We summarize biological aspects and discuss mechanisms of resistance to anti-angiogenic therapy and their implications for treatment selection.
Keywords: Algorithms; carcinoma; molecular targeted therapy; renal cell.
Conflict of interest statement
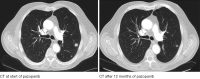
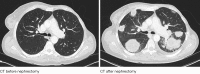
Figures

References
-
- McKibbin T, Frei CR, Greene RE, et al. Disparities in the use of chemotherapy and monoclonal antibody therapy for elderly advanced colorectal cancer patients in the community oncology setting. Oncologist 2008;13:876-85. - PubMed
-
- Gronlund B, Høgdall C, Christensen IJ, et al. Is stabilization of disease a useful indicator for survival in second-line treatment of ovarian carcinoma pre-treated with Paclitaxel-Platinum? Gynecol Oncol 2004;94:409-15. - PubMed
-
- Koopman M, Antonini NF, Douma J, et al. Randomised study of sequential versus combination chemotherapy with capecitabine, irinotecan and oxaliplatin in advanced colorectal cancer, an interim safety analysis. A Dutch Colorectal Cancer Group (DCCG) phase III study. Ann Oncol 2006;17:1523-8. - PubMed
-
- Seymour MT, Maughan TS, Ledermann JA, et al. Different strategies of sequential and combination chemotherapy for patients with poor prognosis advanced colorectal cancer (MRC FOCUS): a randomised controlled trial. Lancet 2007; 370:143-52. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
